[This is an entry to the 2019 Adversarial Collaboration Contest by the delightfully-pseudonymous Adrian Liberman and Calvin Reese.]
About the Authors: Adrian Liberman is currently a PhD student in biology at a university in the mid-Atlantic. He previously worked at the National Institute of Aging and remains actively interested in gerontology and the biological study of aging. Calvin Reese is an author with a BS in Biology. He has always been interested in the possibility of life extension by calorie restriction. Recently, he has reexamined the subject after undertaking a series of intermittent fasts for weight loss reasons. Calvin believes CR extends life; Adrian has long been skeptical.
Introduction: Is food making us old?
We all agree that food is delicious, and we also all agree that too much food is bad for us, but exactly how bad is it? Various academics have proposed that too much food actually accelerates the aging process, and reducing our food intake via calorie restriction (CR) is one of the most accessible and available methods of extending human life. While billionaires pump vast fortunes into increasingly far-fetched stem cell treatments and consciousness transfers, CR advocates contend that they can get a 10-20% increase in their natural lifespans simply by eating a little less. If true, CR raises a question of enormous significance to gerontology and the science of aging: are our diets aging us one calorie at a time? And if so, can we stop it?
Calorie Restriction (CR) and Intermittent Fasting (IF) advocates generally claim that CR will extend your lifespan and prevent various diseases, and that IF is an effective form of CR. Evidencing these claims are animal studies in yeast, worms, flies, mice, and monkeys as well as indirect evidence from humans. A variety of biochemical studies have been performed, and a host of theoretical literature generally claims that the underlying mechanisms for the effect are the IGF axis affecting sugar metabolism, DNA damage mechanisms dealing with free radical formation, and inflammation modulation.
My position (Adrian) is that for the average individual reading this article, CR and IF are not generally worth the effort, because the individual will be exposed to non-trivial risks and the benefits will be minute.
My position (Calvin), is that there exists some amount of food, on average, that will produce an optimal human lifespan and that the average person could significantly extend their life by moderately reducing their calorie intake.
Calorie Restriction (CR) is a term coined to describe a series of experiments that have been conducted over the course of more than a century, demonstrating that various animals kept in laboratory conditions generally survive longer if fed diets that are ‘restricted’. This effect has been observed in bacteria, yeast, worms, fruit flies, mice, and arguably monkeys, so it appears to span every domain of life. (40)
On the other hand, population-level studies say that the lowest observed mortality in western populations occurs at a BMI of ~25. (4)(5) This appears to be a paradoxical result, since a BMI of 25 generally results from a diet that isn’t particularly “restricted”. Why would we observe a lower mortality in lab animals when they are undergoing perpetual mild starvation, but a higher mortality when this happens in humans?
And by extension, should you, the reader, adopt a calorie-restricted diet, or an NIH-approved 25 BMI diet?
Semantics: Calorie Restriction versus just not overeating
First things first. Let’s define some very strict terminology. This will become important later:
Kinds of diets:
• Ad libitum Diet (ad-lib) – at one’s pleasure, or as much food as you would like.
• Ad-lib Calorie Restriction (Ad-lib CR) – a diet that contains fewer (for example 30% fewer) calories than your diet would if you were eating an ad-libitum diet
• Normative Diet (ND) – a diet that is balanced and prevents the onset of obesity. The general analog to USDA’s 2000 calorie diet. (Our term and not in common usage)
• Normative Diet Calorie Restriction (nd CR) – a diet that is balanced, but contains fewer calories than a normative diet (for example 30% fewer)
Once we establish these definitions, the kinds of claims that proponents or opponents can make expand into the following:
Claim 0: You, the reader, should adopt an ad-lib diet
(Nobody claims this, put the chips down!) (null hypothesis)
Claim 1: You, the reader, should adopt an ad-lib CR diet
Claim 2: You, the reader, should adopt a Normative Diet
Claim 3: You, the reader, should adopt an nd-CR diet
For the purposes of this article, we’ll be assuming that you, the reader, are an average American of indeterminate sex.
First the claims of CR in detail:
Animal Studies and the NIA Interventions
CR relative to ad-lib leads to improved metabolic function in the animals studied, generally because they do not become obese. On the other hand CR relative to a normative diet leads to unfavorable/negative outcomes including a decrease in fertility, altered mental states, and muscle wasting, but also a significantly increased lifespan.
A Calorie Restriction experiment goes like this: Take some animals and first establish the amount of nutrients they consume “normally”, or under optimal growth conditions. After this, take half of those animals and provide them access to all the micronutrients and amino acids they want, but restrict their access to raw energy in the form of fats or carbohydrates to X% of their normal intake.
Where does this lead to increased lifespans? Let’s start at the beginning! The beginning of time:
CR dramatically extends the lifespan of yeast. (39) Yeast longevity is difficult to quantify, but experiments have suggested 75% CR in yeast (done by decreasing glucose concentrations in yeast media from 2% to 0.5%) extends yeast longevity by a factor of 3. (39)(40). Single-celled organism lifespan is a goofy term, but it can be measured both directly by looking under a microscope at cells and keeping track of when they kick it, and indirectly by comparing the steady population of cells to how often they divide.
Nematodes (worms) exposed to 50-75% CR experience a 2-3 fold increase in longevity. CR in Drosophila melanogaster produces between 30% and 100% increases in the observed lifespan of the flies. (40)(41) Many hypotheses have been proposed to explain why CR produces such marked increases in the lifespan of worms, flies, and yeast, but most notable is the behavior of the SIRT-family genes. (71) Unfortunately the SIRT mutation data is difficult to interpret due to the relatively central role SIRT genes tend to play to the functioning of a diverse array of cells, but some theories are more convincing than others. More on this later. Of course, in general, the problem with studies like these is that establishing what constitutes a normative diet in organisms like these is somewhat subjective. Is there such a thing as an obese yeast cell? On the other hand, a normative diet for rodents is pretty easy to establish.
Numerous studies and overwhelming evidence show that CR significantly extends the lifespan of rats. Rodent CR studies suggesting CR extends lifespan go back to the 1930s and 1940s (26) (30). The earliest rodent CR studies focused on longevity with modern approaches and methods were conducted in the 1980s. Pugh et. al in 1992 (27), Yu et. al in 1985 (28), and Weindruch and Walford in 1982 (29), all subjected rats to 40% CR diets compared to ad lib baselines and found between 10 and 20% increases in longevity as compared to normative diets. Similarly, 1986 Weindruch is a gold standard mouse CR study. Here we see that even fairly aggressive ndCR produces extended average and maximum lifespans, both relative to ad-lib and relative to a normative diet, with a normative diet extending lifespan 20% relative to ad-lib, and 40% ndCR extending lifespan another 30% on top of that. (Context: think big fat guy keeling over from a heart attack at 50, vs everyone’s stereotypical tiny Chinese grandma, who spends her 100th birthday stubbornly refusing to reveal the location of her phylactery)
Reported health benefits of CR in rodents include reduced cancer risk in p53-deficient mice (33), increased proteasome activity (34) in mice and rats, improved cognition (32)(35), reduced oxidative stress and NF-kB signaling (36), and various other health benefits. Several studies, including Park et. al. and Pires et. al. have observed dramatic changes in insulin signaling and serum glucose levels (32)(37)(38), which has been of particular interest to gerontology researchers. IGF signaling has been proposed as one of the mechanisms by which CR improves health outcomes (15).
So far this is a pretty strong story. What’s the sketch factor of this evidence? Well… Some rat and mouse strains responded to CR better than others and methodology varied widely between rodent trials, making them difficult to compare (32). The only study performed on wild-caught mice that aren’t buried under a mountain of genetic defects to shame the Habsburgs had a negative outcome. As a model for aging, mice are also slightly suspect because unlike most mammals, they are globally telomerase-positive, which means that the effects of mutational accumulation on their soma don’t directly translate to other mammals, since oxidative damage has a large interplay with telomeric senescence.
Overall, however, we agree that mouse lifespans are significantly extended by CR. But…
As the organism becomes larger and more complex, the beneficial effects of CR on lifespan appear to taper off (40). 25% nd CR extends dog lifespan 25% (66). Why isn’t more aggressive CR investigated in dogs? Larger animals don’t tolerate aggressive caloric restriction well, 40% caloric restriction would likely kill most dogs. It seems that brains are probably to blame, because the metabolic rate of the brain is generally not significantly regulated (70). If you run out of energy for running your brain cells, you’re done for. The larger the proportion of your metabolism dedicated to maintaining brain function, the less CR you can tolerate.
So, given that trend, the most relevant information for humans should probably come from monkeys. (#freescopes)
Unfortunately, monkeys live a long time, so studies of monkey aging are measured in many decades and cost millions and millions of dollars. The National Institute on Aging and the University of Wisconsin have taken on the task and subjected ~40 rhesus monkeys each to 30% CR. In the WNPRC study, CR was versus an ad lib baseline and in the NIA study CR was relative to a standardized diet designed to prevent obesity. (1) (3)
Rhesus monkeys have a maximum lifespan of ~40 years, and after 30, the WNPRC study reported only 13% of the CR group had died of age-related causes, whereas 37% of the control group had died. (1) The authors write “CR reduced the incidence of diabetes, cancer, cardiovascular disease, and brain atrophy.” (1) In 2017, 10 of the WNPRC CR group animals remained alive compared to 3 animals in the control group. (31) In a monument to absurd timelines, the WNPRC study is not yet over, as many CR monkeys survive in 2019. No mean lifespans are established.
Here we hit our first snag. In complete contrast to the WNPRC study, the NIA rhesus monkey study used a standardized diet designed to prevent obesity for the control group. The NIA study found no increase in survivability among the CR group. (3) What gives?
If we were being charitable, we would say that monkey studies are ridiculously hard to power properly. With only 86 monkeys present in the NIA study, the negative observation could easily have been a product of chance that was inadequately represented by the reported p-values, and we should defer to priors based on other mammals. Anecdotally, and though the NIA paper would never admit to this, the NIH sometimes staffs monkey studies of this kind with “leftover” monkeys from clinical trials that may have mysterious medical conditions that are not obvious to the naked eye but were caused by drug trials, experimental surgical procedures, etc.
If we wanted to be less charitable, we would look at the fact that the WNPRC study pulled shit like this:
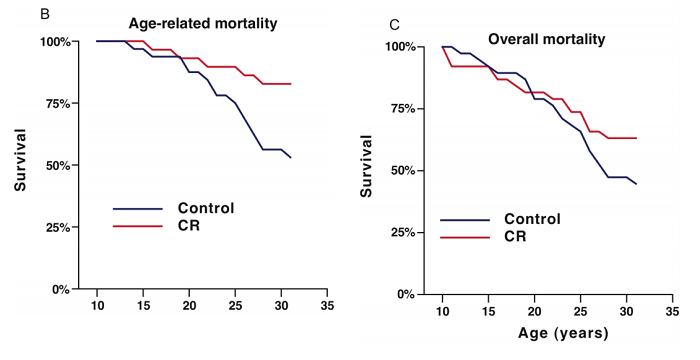
…and point out that aging is almost by definition a process that impacts the survival rate relative to any injury, so trying to disentangle “age related” mortality from regular mortality is bad and wrong.
The most significant difference between these studies is the use of adCR (Wisconsin) vs ndCR (NIA). It’s no surprise that monkeys that are on a controlled diet are healthier and live longer than monkeys that are fed an ad-libitum diet. In mice, this is a commonly known problem for control populations, and monkeys are no different (72). The balance of evidence probably tilts against the idea that CR is effective in monkeys, however our priors that ndCR should be effective are fairly strong, so for now let’s assume that it is.
Adrian concedes that a 2017 statistical analysis of both studies by the University of Alabama at Birmingham in cooperation with the authors of the original studies determined that CR decreased mortality in rhesus monkeys (31), but the study is presented under protest because combining ndCR and adCR in the same analysis is inadvisable, disingenuous, probably illegal, and was the direct cause of the sinking of the Lusitania.
The gold standard of CR studies in animals with respect to human health would be studies that occur in higher animals (mice, dogs, monkeys) and perform CR relative to a normative diet. On the balance, evidence that ndCR extends lifespan in higher animals is fairly strong. The monkey problem, however, is pretty bad, and we remain skeptical of the strength of CR in humans.
Despite these objections, we agree that CR promotes longevity and reduces all-cause mortality of animal model organisms and that this finding supports the view that CR probably increases the lifespans of humans in an ideal scenario.
So… what’s up with that? Do the above findings mean that we need to radically reinterpret what being overweight or obese means? Are we just WILDLY overestimating the “healthy” weight for all these animals?
In a word, no. Animals are clearly not adapted to undergoing caloric restriction this severe. How do we know that? Easy. The animals in most of the experiments above, under fasting conditions, are driven sterile. From an evolutionary standpoint, it’s safe to assume that being sterile is not an adaptive trait, so clearly most animals have not evolved to operate at levels of caloric restriction this severe on a routine basis. That’s one of the simpler distinctions between a “healthy weight”, obesity, and being in a starvation regime. Still, clearly animals live longer when they are starving, so why?
Proposed Mechanism by Which Food Makes You Old (and how calorie restriction stops it)
First an interlude: aging is one of the last frontiers in biology where major theories still compete on an even footing to explain a basic and universal process. The simplest definition of aging is the observation that past a certain point in an animal’s life, the likelihood that it will die doubles every period X, where X is different for different species. This observation is stunningly universal. It’s also important to note that the likelihood of death from almost any type of injury increases over time, so aging is not just the idea that cancer is more frequent when you’re 50 than when you’re 20, but the idea that almost all diseases are more frequent in older individuals, and dying from almost any injury is more likely when you are old than young (78, Arkin).
Covering the slapfight over the specific mechanism involved fully is beyond the scope of this paper, especially because there are no conclusive answers to any of your questions, but briefly these are the major aspects of biology that change with age, drawing directly from “The Hallmarks of Aging” Lopez-Otin et al. (44).
1) genomic instability, such as replication errors, mutations, DS breaks, and crosslinking;
2) telomere attrition, including damage to the telomeres that does not result in telomere shortening;
3) epigenetic alterations, including changes in methylation patterns, histone modifications, and chromatin remodeling. This generally also leads to deregulated/erroneous gene expression;
4) loss of proteostasis, which is characterized by protein denaturation or unfolding and the accumulation of waste products your body cannot break down;
5) deregulated nutrient sensing, causing both the cell and the body to become less responsive to nutrients;
6) mitochondrial dysfunction, which is thought to be particularly central to the relationship between CR, obesity, and aging because mitochondria process glucose and create reactive oxidative species (ROS);
7) senescense or quiescence, the cessation of the cell cycle; and
8) stem cell exhaustion, which slows or halts renewal of virtually all tissues and cell types.
These are specific, measurable instances of the general breakdown we associate with aging chosen for their correlation with chronological and apparent age. None of them has been decisively established as the actual cause of aging as we understand it, though all are understood to contribute to aging at the cellular and tissue level, which is the general breakdown and cessation of cell functionality (44).
Gerontologists love to whip these hallmarks out, but in reality most of them are interrelated in some way, so establishing which ones of them are merely the byproducts of other is extremely difficult. Ex: mitochondrial dysfunction increases the rate at which oxygen radicals are produced leading to greater genome instability and telomere attrition. Stem cell exhaustion probably arises from genomic instability or nutrient sensing deregulation, but on the other hand it can lead to senescence in the tissues as replacement of dying cells slows down. It’s all an ouroborosian mess. (44)
Almost all of the hallmarks have been shown to be impacted by food intake. Obesity has been decisively implicated in causing genomic instability in model animals, but evidence is lacking in humans (45). Crucially, the CR-ROS hypothesis that excessive eating, causing both oxidative damage from digestion and metabolism of glucose and obesity – thus linking obesity and ROS damage together, remains poorly supported in humans (45)(46). Obese adult individuals suffer from greater telomere attrition than non-obese individuals and have shorter telomeres (47). Obese individuals have profoundly different epigenetics than non-obese individuals, which is not surprising, since insulin expression profiles and fat metabolism are well-established as being changed by, and changing, epigenetics (46)(48)(49). In fact, epigenetic changes in insulin expression due to a high-fat diet may be heritable (48). Obese individuals, and particularly diabetes patients, have markedly different DNA methylation patterns from non-obese persons and modified chromatin structure (49) Altered chromatin density due to obesity has been observed in rodents and implicated in the onset of dementia, which is commonly associated with old age (50)(51). Numerous proteasome dysfunctions and significant protein misfolding have been observed in obese rodent and human subjects (46). In addition to creating insulin resistance, human adipose tissue creates a pro-inflammatory environment and significantly alters cell response to NF-kB and inflammatory cytokines (52). Obesity has been specifically implicated in ROS damage to mitochondria, mitochondrial dysfunction, reduced mitochondrial fission, and further elevated ROS production by the Krebs cycle (53). Furthermore, adipose tissue and obesity apparently promote senescence (54). Obese mice exhibit dramatically increased rates of T-cell senescence (55). Finally, obesity results in far greater stem cell exhaustion and quiescence (56). Adipose tissue stem cells, hematopoetic stem cells (HSCs), bone marrow, and other stem cell reserviors have all demonstrated higher rates of quiescence in obese populations of rodents and humans (46).
So, we have covered all eight biomolecular cell and tissue-level hallmarks. Obesity does, in fact, make us older. And we are pretty sure CR makes most animals younger. That is an excellent clue as to how the two are related, and which of the mechanisms might be the most important.
Your body usually converts sugar into mechanical or chemical energy by using a complicated daisychain of a couple dozen proteins called the Electron Transport Chain (ETC). The ETC is so called because each protein in it contains an electrically charged amino acid that is highly reactive due to holding on to loose electrons that came from sugar. The ETC takes electrons obtained from sugar, and attempts to stick them onto molecules of oxygen, converting it into water. Unfortunately, this process occasionally fucks up, and instead of getting nice benign water, the oxygen becomes radicalized, gets an Al-Qaeda franchise, and becomes either Hydrogen Peroxide or the Superoxide anion. Both of these are comically reactive molecules, and if they happen to diffuse out of the mitochondrion, they can react with pretty much anything and cause damage. If they react with your DNA, you’re in trouble, because oxidative damage to your DNA produces mutations. Similarly, the oxidation of fats results in chemicals that cause inflammation, and the oxidation of proteins can result in byproducts that your body can’t break down.
From here comes the Rate of Living Theory: an animal can consume only a specific and finite number of calories in its lifetime, because after that point the damage induced by said calories becomes fatal.
Essentially, for a given cellular architecture, so many calories in results in so many molecules of peroxide and superoxide out, and so many mutation events in DNA, oxidized proteins, and rancid fats. Inside you there are populations of cells that can tolerate only a finite amount of oxidation events, namely heart muscle fiber nuclei, neurons, and long-term stem cells like marrow and fibroblast stem cells. For these tissues, the DNA you have is the DNA you get, and you can only expose it to so much chemical damage before it stops functioning. Similarly the accumulation of other byproducts would be fatal.
While there is some slight debate about this, the balance of evidence is that CR slows down the metabolism of most tissues relative to their quantity (74,75), so it should definitely allow you to stretch your “mutation budget” over a longer timeframe. This theory predicts that you can extend your remaining life by X% by cutting X% of calories out of your daily food intake. Napkin math tells us that this basically tracks with the results we saw above, and the limitation on this is obviously starving to death.
Hopefully at this point we have exhaustively convinced you that Calorie Restriction definitely works in small animals, probably due to inhibiting oxidant production, and it might work in humans, though the theoretical backing for this is dicier.
So we return to our claims. Should you eat an ndCR diet?
First we have to take care of Chesterton’s Fence.
Population Aging Studies and Human Trials
Why is it commonly recommended to consume ~2000 calories per day? And where does this number come from anyway?
Dietary guidelines approach the question of how much food you can eat epidemiologically, by asking what kinds of mortalities are associated with various food intakes.
A meta-analysis of large-scale population studies concludes that a BMI of roughly 24, considered to be “normal” weight, is associated with the minimum long-term all-cause mortality. (5) (4), and once BMI exceeds 30, all-cause mortality begins to sharply increase. (5) (4) A CDC estimate for the 2015-2016 period found that 39.8% of adults were obese, and it is not unreasonable to assume that the average American BMI is roughly 30 based on huge body of evidence. The interesting aspect of these studies lies in the fact that BMIs below 25 are likewise associated with increased mortality (5)(4). Direct observational data of western populations is nearly unanimous in showing that BMIs below 25 are correlated with bad health outcomes.
So here we meet the fundamental paradox: Why is it that we can observe lab animals living longer in the face of CR, but when we observe humans, we generally find an association between mortality and low body weight?
Epidemiologic studies on low BMIs in western populations become a little hazier. There is fairly wide agreement that BMIs below 25 correlate with lower survival, but not usually a clear claim as to why. BMIs below 25 generally appear to correlate with smoking. Excluding the effects of smoking, people with BMIs below 25 still appear to have increased all-cause mortality. This could be from correlations to weight loss from cancer and metabolic diseases, tuberculosis, or something similar. While the effects of obesity on risk of mortality are very clear, the effects of being underweight and starvation on health are obviously the source of our core paradox (4).
Even studies that attempt to exclude acute illnesses that commonly induce weightloss seem to find higher rates of death among men that are underweight across a wide range of causes. Most troubling is the fact that an under-consumption of calories has a documented effect of suppressing wound healing and immune response, and worsening the progression of infectious diseases (67-69). From this standpoint, it’s altogether unclear whether the low weights observed in these studies are induced by the diseases in question, or whether diseases are simply more prevalent in people with BMIs below 25.
Calvin’s hypothesis, though unsupported by evidence, is that no one is actually on a CR diet.
A CR diet is reduced calorie intake without malnutrition. Some of the people with BMIs below 25 may have an underlying health issue, such as HIV, which is causing their BMI to drop or mortality to rise, but I speculate that most people with a BMI below 25 are suffering from malnutrition, which is causing the increased mortality observed in persons with BMI below 24-25. Sarcopenia in the elderly is another possibility, though age-adjusted studies appear to refute this hypothesis (4).
We can attempt to look at other cultures to see if lower caloric consumption has similar effects there.
Gerontologists and anthropologists have observed that the longest-lived national and ethnic groupings of humans tend to eat the least. Japan has long had the highest life expectancy of any developed country (20). Japanese life expectancy at birth stood at 87.17 years in 2016, as compared to 81.40 years for the United States and 84.43 being the average of 18 high-income countries (20). Japanese are estimated to eat 23% less than Americans (21).
However, this finding is purely correlational in nature. Although reverse causation – that people eat less because they live longer – can be ruled out, the correlation could be coincidental, pleiotropic, or for genetic rather than dietary reasons. The FOXO3A gene, rather than CR, has been proposed as the reason for variations in longevity between ethnic populations (17). Several other genes, like APOE and CETP, have been suggested as alternative genetic causes of these ethnic longevity differences (18). Gerontologists have aggressively suggested that smaller humans tend to live longer (4) (13) (14), and that members of ethnic or national groups, such as the Japanese, who live longer tend to be physically smaller, and centenarians within this ethnic group tend to be smaller than those who live shorter lives (13). One proposed reason that smaller individuals and ethnic populations tend to live longer is lower levels of GH and IGF-1 due to genetic factors (15). As has been discussed, IGF-1 is associated with increased mortality due to various illnesses and also makes the individual physically larger. CR advocates have equally argued that social, environmental, and economic factors causing CR in these population groups cause the drop in GH and IGF-1, increasing longevity in these groups. A Washington University study of the effects of CR on a group of humans on a long-term diet of 1800 kcal per day versus an experimental group ingesting 2500 kcal per day found no decrease in serum IGF levels from baseline unless protein intake was also restricted (16). This result is at odds with rodent studies, which showed decreases in IGF-1 concentrations in CR subjects versus those on normative diets (16). Other results contradict these findings. A Tufts 30% CR trial among mildly overweight (BMI 25-29.9) young adults showed significant decreases in serum insulin concentrations, contradicting the Washington University results (25). Many, many other human trials have produced contradictory results in CR trials, with the severity of the CR, weight, and age of the patients, as well as compliance with CR, having been variously proposed as explanations (21).
There is also compelling evidence in favor of CR and dietary explanations as the cause of longevity in particular populations. For example, the ethnically distinct Okinawans were for generations the Japanese ethnic group with the greatest longevity; the island has 4-5 times the centenarians per capita of any industrialized country (21). Little racial admixture has occurred on Okinawa, but as the Okinawan diet has westernized, the Okinawans have lost their longevity advantage, with Okinawan longevity dropping below the rest of Japan in 2005 (19). Older Okinawans, who continue to eat CR and protein-restricted traditional diets, have greater longevity compared to Japanese populations of equal age. For Okinawans age 60-64, all-cause mortality was half that of Japanese persons of equal age (22). Evidence from natural “experiments” during mandatory rationing and food shortages also supports the CR hypothesis. Involuntary food rationing during the World Wars also paradoxically increased lifespan. WWI-era rationing in Denmark resulting in a 34% drop in mortality over a two year period (21)(23). Similar rationing in Oslo during WWII, thought to be equivalent to 20% CR, resulted in a 30% drop in mortality (24).
The longest lived humans tend to eat the least. Though causation – whether CR causes decreased IGF levels or decreased IGF levels cause CR – cannot be definitively established, the conclusion, supported by rodent studies and defensible from surveys of humans, that CR causes a drop in IGF levels remains compelling.
So, should you, mean American Reader with 1.5 X chromosomes, adopt a CR diet? WHAT COULD GO WRONG?
Acute Risk Factors of Caloric Restriction
A good first place to look is the Minnesota Starvation Experiment. Conducted around 1944 on 32 volunteer males, the object of the experiment was to subject humans to a 25% calorie-restricted diet that simulates 6 months of famine and observe the effects. Diet was strictly controlled by housing subjects in a special dormitory and supervising them during their time outside of it. Despite the simulated famine conditions, diets were formulated to ensure that subjects received daily minimum intakes of important vitamins and minerals
An exhaustive description of the effects can be found in The Biology of Human Starvation, by Keys, but let’s go through the highlights. (70, not available online but try to find it at a library. Equal parts fascinatingly and grim, but occasionally also very funny)
First the physical: subjects of the experiment underwent a substantial drop in bodyweight (duh) over the first few weeks, after which their weight stabilized at 75% of their original bodyweight, as was the design of the experiment. In terms of physical strength and work capacity, subjects experienced loss of motor coordination, loss of strength, and a devastating negative impact on their endurance. Counts of all blood cells were down across the board per unit volume of blood, and in absolute terms. Subjects had a lowered basal metabolic rate, and an observed drop in surface but not core body temperature. Severe caloric restriction (15%+) also leads to a sharp fall in fertility for both men and women. Women undergoing caloric restriction may stop menstruating. (not observed in the Minnesota study, which was all-male) Finally, a starvation state can lead to bradycardia, starvation edema, fainting, and looking like a cartoon skeleton (70)(77).
Psychologically, the first and most significant observed effect was a severe preoccupation with food, and a permanent feeling of being cold. The latter was both a true sensory perception (skin temperatures of subjects were lower than normal) and a subjective sensation. Additionally, subjects experienced a loss of sexual interest. The subjects were capable of achieving erections physiologically, but not psychologically. Subjects also felt a loss of motivation to engage in self-improvement or social activities. By month 6 of semi-starvation, more than half of all subjects were routinely failing to complete basic maintenance tasks such as cleaning, and most had dropped out of university classes they were initially attending. After subjects were released from the experiment, several developed eating disorders, and fasting in general can induce eating disorders where none were otherwise present, both binging and anorexia.
Advocates of intermittent fasting, which is a diet that is much easier to maintain, often say that you eventually adapt to the feeling of acute, distracting hunger that strikes you when you are on a short (eg 1 or 2 day) fast (Original research). No such adaptation was observed for the subjects of the Minnesota experiment, and if anything morale of the subjects deteriorated steadily throughout all 6 months of the experiment. In principle, we need not assume that you cannot psychologically adapt to permanent starvation conditions, however mice that undergo lifetime caloric restriction do show a permanently depressed level of motility, eg they just hate moving around.
Evidence from other sources also indicates that lowered caloric intake leads to worsened progression of infectious diseases and slowed wound healing. This point may initially appear controversial because some studies indicate faster and greater response to mitogens, but studies using live pathogens prove this point fairly conclusively. Crucially, this aspect of starvation biology is one that would not be revealed in CR Mouse studies. Lab mice are generally kept in aluminum shoeboxes in a strictly sterile environment most of their lives. They have little opportunity to suffer injuries or heal from the same, and rarely experience infections. If they do, they are quite likely to die and be excluded from analysis. Unless you happen to live in a sterile aluminum shoebox as well, consider this as you interpret CR studies (67-69).
How does this square with the smaller diets in East Asia? Okinawans reportedly ate a diet that was at a similar level of caloric restriction (20% relative to western ND) to the Minnesota experiment, however obviously the entire island wasn’t driven sterile or languid. Note however, that this difference is reported only in absolute terms, not as calories per unit of body weight. Factor 1 is probably stature. Long-term caloric shortages have been shown in both mice and humans to lead to shorter stature and smaller size. If you were reared from youth to eat a relatively smaller diet, your long-term caloric requirements could probably be lowered somewhat. This is borne out in the average heights and weights of Okinawans of this time period, often reported to be less than 5 feet tall. This also squares with studies on longevity between different mouse strains, which routinely report that smaller overall stature (or length) has a positive correlation to longevity.
Factor 2 may be local weather conditions (Okinawa is sub-tropical) Temperatures have an impact on the ability to tolerate a restricted diet long-term. One notable result observed in investigations of Okinawans is the higher thermogenesis and lower oxygen consumption in Japanese and Okinawan mitochondrial haplogroups, suggesting both lower generation of ROS, and likely an ability to tolerate lower caloric consumption while maintaining adequate body temperature (76), which is an important aspect of determining basal metabolic rate. The BMR of Japanese people and Okinawans can be considered as lower or higher depending on whether it is measured through thermogenesis or oxygen consumption, which measure subtly different things.
This discrepancy suggests to us that even in the event that you are able to maintain a traditional Okinawan diet, if you were reared in America, it’s quite possible that you would have the ol’ Minnesota Boner Downer experience attempting to do so.
The outcomes of a lesser caloric restriction would be easier to tolerate for the average westerner, but will also be less effective. Whether there exists a sweet spot in which you are calorically restricted, but don’t hate life and can lift a broom is probably a subjective judgement.
Overall Conclusions
None of the evidence in favor of CR is indisputable. CR-ROS, the hypothesis that calorie restriction reduces oxidative radicals, remains compelling, but direct evidence in humans is lacking. Numerous model animal studies have shown a link between CR, reduced mortality, and life extension. Population studies support the CR hypothesis, but the effects of CR cannot be easily disentangled from genetic, social, environmental, or non-CR dietary factors. CR experiments in humans and rhesus monkeys produced contradictory results, and in some cases the tradeoffs between early and late mortality are a judgment call. Progerias in the obese and biomolecular evidence of cellular and tissue-level anti-aging effects of CR remain the strongest evidence for CR’s potential to extend human life.
We, the authors, conclude that the evidence as it stands weakly supports the conclusion that CR modestly extends human life. We expect that an individual engaging in 20-30% CR versus a normative, non-obesogenic diet without malnutrition might enjoy a 10%-20% increase in longevity. A 10%-15% CR relative to a normative diet may increase lifespan by perhaps 5-10%.
As with all good science, this conclusion raises still more questions. 20-30% CR might result in a 10% increase in longevity, but is that worth it? Calvin, one of the authors, is a practicing intermittent faster and can testify that CR and IF are unpleasant, difficult, and sometimes painful.
Scientific investigation adds another layer to this subjective answer: starvation conditions are likely to expose you to infections of greater severity, potential sterility, negative impacts on your physical abilities, and subjective but significant impacts on your psychological state, including motivation, attention, and libido.
The field of gerontology and the general study of aging continues to lurch forward – not at the pace we want it to, necessarily, but it’s still developing anyway. New drugs and treatments, including stem cell activators like GDF11, senolytic drugs, and anti-inflammatory interventions may be able to make many of the benefits of CR redundant in the relatively near future (we hope).
While CR would probably extend your life, we, the authors, don’t advocate it. The risks and miseries aren’t trivial and you probably have to go to work in order to exchange money for goods and services.
Claim 2: You, the reader, should adopt a Normative Diet
True (and basically the same as an adCR diet)
Claim 3: You, the reader, should adopt an nd-CR diet
Debatable, but no.
If you are interested in the best current options for life extension, you should consider a long-term aspirin regimen, maintaining a healthy body weight, and building a nuclear bunker in your backyard.
All the best,
–Calvin Reese, Adrian Liberman
PS(A): PLEASE NOTE: If you are over 75 years old, do not attempt Calorie Restriction. If your grandma is over 75 years old, go to her house and pour soup into her until she is overweight. This is not a joke and is entirely serious advice. Among the very elderly, being overweight serves as a protective factor that mitigates the dangers of death due to traumatic injury. The dangers of heart attacks and diabetes associated with excess weight are less than the dangers associated with sarcopenia and cachexia. Elderly people usually experience a loss of strength in esophageal muscles and for them swallowing food becomes more difficult, leading to a vicious cycle of muscle weakness and weight loss. If you have an elderly relative, please make sure they’re eating enough.
PPS: Studies excluded from this review: CALERIE: conformity to study protocol was terrible, duration too short, and they took overweight people and got them to baseline. Total tripe on a bike. CRONies: lmfao. Be skeptical of studies claiming to observe DR in a human population. Talking a large group of people into actually following a strict DR regimen long-term is borderline impossible because it fucking sucks. Sample was also self-selected.
Bibliography
1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2812811/ – Wisconsin Monkeys
2. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC345016/ -84 adiposity
3. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3832985/ – NIA Monkeys
4. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4115619/ – Mortality data
5. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2662372/ – BMI meta-analysis
6. https://qz.com/255210/turns-out-the-way-americans-measure-healthy-weight-is-totally-wrong/ – media report on American obesity (for estimating average BMI)
7. https://www.nbcnews.com/healthmain/real-shape-american-man-dudes-youre-porky-8C11394082 – media report on American obesity (for estimating average BMI)
8. https://www.theatlantic.com/health/archive/2013/10/this-is-the-average-mans-body/280194/ – media report on American obesity (for estimating average BMI)
9. https://www.cdc.gov/nchs/data/hestat/obesity_adult_13_14/obesity_adult_13_14.pdf – NIH obesity 2013-2014
10. https://www.cdc.gov/nchs/data/databriefs/db288.pdf – CDC obesity 2015-2016
11. Dixon, John B. “The effect of obesity on health outcomes.” Molecular and cellular endocrinology 316, no. 2 (2010): 104-108. https://doi.org/10.1016/j.mce.2009.07.008
12. https://www.ahajournals.org/doi/full/10.1161/ATVBAHA.111.241927 – biomolecular consequences of obesity
13. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3354762/ – various characteristics of centarians
14. https://www.ncbi.nlm.nih.gov/pubmed/12208237/
15. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3893695/ – effects of IGF and GH on body size, longevity, and mortality
16. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2673798/ – effects of CR and protein restriction on IGF serum levels
17. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4707876/#MXS013C74 – FOXO3A variation and longevity
18. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4296168/ – APOE variation and longevity, supports FOXO3, refutes others
19. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3362219/ – Okinawan longevity
20. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6092679/ – global life expectancy trends by country
21. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5315691/ – lots of stuff, general review of CR
22. https://www.ncbi.nlm.nih.gov/pubmed/16810568 – calorie restriction in Okinawa
23. https://jamanetwork.com/journals/jama/article-abstract/223580 – involuntary rationing in Denmark during WWI
24. https://www.ncbi.nlm.nih.gov/pubmed/14795790 – involuntary rationing in Norway during WWII
25. https://www.ncbi.nlm.nih.gov/pubmed/17413101 – Tufts CR study that showed decreased insulin levels in overweight subjects.
26. https://academic.oup.com/jn/article-abstract/21/1/45/4725572 – 1941 rodent CR study
27. https://www.ncbi.nlm.nih.gov/pubmed/10197641/ – Pugh et al. 40% rodent CR study
28. https://www.ncbi.nlm.nih.gov/pubmed/4056321/ – Yu et. al 40% rodent CR study
29. https://www.ncbi.nlm.nih.gov/pubmed/7063854/ – Weindruch and Walford rodent CR study
30. https://www.ncbi.nlm.nih.gov/pubmed/2520283 – 1935 McCay rodent CR study
31. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5247583/ – 2017 combined Rhesus monkey study
32. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5565679/ – review of rodent CR studies
33. https://www.ncbi.nlm.nih.gov/pubmed/12016155/ – reduced cancer risk in p53 deficient mice
34. https://www.ncbi.nlm.nih.gov/pubmed/17460208/ – increase proteasome activity in CR mice and rats
35. https://www.ncbi.nlm.nih.gov/pubmed/18002475/ – improved cognition in CR mice and rats
36. https://www.ncbi.nlm.nih.gov/pubmed/19199090/ – anti-inflammatory effects of CR in rodents
37. https://www.ncbi.nlm.nih.gov/pubmed/16920310/ – improved glucose tolerance and lower insulin levels in CR rodents
38. https://www.ncbi.nlm.nih.gov/pubmed/24844367/ – drop in serum insulin in CR rodents
39. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3696189/ – review on budding yeast longevity
40. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3607354/ – review on longevity in general
41. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1857724/ – Drosphila CR review
42. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3181168/ – Progeria rapamycin
43. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6509231/ – “Obesity May Accelerate the Aging Process”
44. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3836174/ – The Hallmarks of Aging
45. https://www.ncbi.nlm.nih.gov/pubmed/30115431/ – genome damage by obesity
46. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6509231/ – obesity/aging review
47. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2805851/ – telomere attrition in the obese
48. https://www.ncbi.nlm.nih.gov/pubmed/21088573/ – nutritional epigenetics
49. https://www.ncbi.nlm.nih.gov/pubmed/24779963/ – methylation patterns and diabetes
50. https://www.ncbi.nlm.nih.gov/pubmed/24154559/ – obese TD2 rat chromatin density
51. https://www.ncbi.nlm.nih.gov/pubmed/25380530/ – more obese rodent chromatin density
52. https://www.ncbi.nlm.nih.gov/pubmed/27503945/ – pro-inflammatory environment of adipose tissue
53. https://www.ncbi.nlm.nih.gov/pubmed/29155300/ – mitochondria ROS/dysfunction in the obese
54. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2941545/ – obesity and senescence review
55. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5127667/ – obesity and senescence in mice
56. https://www.ncbi.nlm.nih.gov/pubmed/22772162/ – stem cell quiescence in adipose tissue
57. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5599616/ – methylation drift in rodents, monkeys
58. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2190719/ – CR’s effect on genomic stability
59. https://www.ncbi.nlm.nih.gov/pubmed/17665967/ – CR promotes autophagy of misfolded proteins in rats
60. https://www.ncbi.nlm.nih.gov/pubmed/30395873/ – CR and cell senescence review
61. https://www.ncbi.nlm.nih.gov/pubmed/25481406/ – CR reduction of stem cell exhaustion
62. https://www.sciencedirect.com/science/article/pii/S1934590912001671 – CR and skeletal muscles, including transplant
63. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4889297/ – SIRT6 and NF-kB
64. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4570809/ – SIRT-1 signaling and CR in rats
65. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3331748/ – AMPK causes insulin sensitivity in CR mice
66. Kealy, Richard D., Dennis F. Lawler, Joan M. Ballam, Sandra L. Mantz, Darryl N. Biery, Elizabeth H. Greeley, George Lust, Mariangela Segre, Gail K. Smith, and Howard D. Stowe. “Effects of diet restriction on life span and age-related changes in dogs.” Journal of the American Veterinary Medical Association 220, no. 9 (2002): 1315-1320. https://admin.avma.org/News/Journals/Collections/Documents/javma_220_9_1315.pdf?7fh285_auid=1555113600043_jueqhhd825meind2et (Dog CR)
67. https://academic.oup.com/biomedgerontology/article/60/6/688/590315 (live infection in CR mice)
68. https://link.springer.com/article/10.1007/s11357-008-9056-1(live infection in CR mice)
69. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3528375/ (wound healing in CR mice)
70. ISBN 978-0816672349, Ancel Keys, Biology of Human Starvation (Minnesota starvation)
71. https://www.sciencedirect.com/science/article/pii/S1043276009000915 (SIRT mutants vs CR)
72. https://www.pnas.org/content/pnas/107/14/6127.full.pdf (Mattson control mice)
73. The Retardation of Aging in Mice by Dietary Restriction: Longevity, Cancer, Immunity and Lifetime Energy Intake1 – Weindruch mouse study, 86
74. https://www.sciencedirect.com/science/article/pii/S0047637407001005 CR metabolic rate 1
75. https://www.sciencedirect.com/science/article/pii/S0047637499000949 CR metabolic rate 2
76. https://jphysiolanthropol.biomedcentral.com/articles/10.1186/1880-6805-31-22 Okinawa thermodynamics
77. https://www.cambridge.org/core/services/aop-cambridge-core/content/view/96E9E7436516D443974E1C6C8859299D/S0029665194000170a.pdf/the-right-weight-body-fat-menarche-and-fertility.pdf Starvation/Fertility women
78. Biology of Aging: Observations and Principles, Arkin, general description of aging theory (doubling mortality rate)
















Coming in late, but this seems so wild that it wrecks my confidence in the whole rest of the piece. The context is “western populations” – is there a missing caveat? Half of Europe has a BMI under 25.
CR diets: you don’t actually live longer, it just feels that way.
Nicely written piece, thank you both.
Unfortunately it was almost completely useless for me because I’m not an average American with 1.5 X chromosomes. To me it seems mad that you say that “ND” is approximately the same as “adCR”. I have a BMI of 18.5 and eat at least 2500kcal per day (which I increase if my BMI drops below 18.5), but I can very easily reduce the amount I eat. When I don’t force myself to eat more than I feel like, I generally eat less than 2000kcal and lose weight. So I was reading this hoping that I’d learn whether I’m right to be stuffing my face with >2500kcal to maintain BMI of 18.5 or whether I should just eat what I feel like and lose weight. But I came away thinking that I should have a BMI of 25 *and* I should eat less than I currently do! I mean fair enough, I’m a big outlier, but I was still a wee bit disappointed to come to such a contradictory conclusion.
(I don’t have any illness or condition that causes weight loss btw, at least not that I’m aware of. I am very fit and healthy, even compared to other mid-twenties men.)
Intentionally haven’t read the comments yet, but will update this if it’s been addressed.
The longevity extension from calorie reduction would fit a clear evolutionary role, and would be easy to test (except for those pesky IRBs). An otherwise procreation-capable person (but especially female) who is experiencing famine is unlikely to birth a child who will live to procreate themselves, but as a direct effect (gestating mother starves from the increased demand without increased supply) and indirect (child starves after birth). But if you can delay procreation until it’s safe to do so you increase the odds that your genes pass to future generations. Test: do people whom experience starvation regain fertility afterwards (weak) and do people who experience extended starvation regain fertility to ages when it is uncommon – do women who starved from 20-50 regain fertility for the following decade (strong)?
On the starvation studies, there’s some key points that were missed, and some anecdata. We have a group engaging in a natural starvation experiment on a regular basis: body sculptors. While they don’t fit into any other common group the way their hobby works is to gain as much muscle mass as possible, consuming tremendous amounts of protein and gaining significant fat as well, followed by extended periods of minimal calorie consumption to remove all of the fat (and water!) they’d gained while maintaining the muscle with small amounts of protein and very mild exercise. These people, and others who use extreme calorie reduction for weight loss, tend to report almost unanimously that it gets much easier after about two weeks of sub-500 calorie diets (and often sub-300 calorie). These diets are often structured as an egg, a can of tuna fish, and celery and water (with occasionally a teaspoon of fish oil), which in conjunction with supplemented micronutrients allow rapid and extreme weight loss (2 pound per week is considered slow under these circumstances) because even though their body is trying to minimize their BMR there’s no way to make up that large of a deficit.
This pattern is also seen in body building groups where someone is exercising vigorously at 1800 calories and unable to lose weight precisely because they aren’t consuming enough calories to raise their BMR. This is likely what plagued the Starvation study – they were consuming too few, and yet not enough calories. People using extremely low calorie diets to lose weight, whether for health reasons or for competition do however report many of the same symptoms – forgetfulness, lack of attention to detail, decreased energy, etc.
With this background I suspect the starvation experiment didn’t go far enough, and since they weren’t testing hormones we don’t know which of those effects were from starvation induced hypogonadism compared to starvation itself. A follow up (again, cursed IRBs) should include weekly hormone panels, a sub group on hormone replacement therapy, and multiple calorie levels. Then we’ll have much better data to make a hypothesis we can then go test.
On low BMI having higher mortality – I’d assume that’s BMI on death, so by definition every eating disease will be reported as having killed a person with a low BMI… which kills that meaning. A person with a BMI of 24 who has a serious infection will often lose 10 or more pounds before it kills her, so unless we can separate by cause of death, co-morbidities, and BMI prior to final decline I’m not sure that really means much. It may be that the ideal human BMI is 20, but that because extra fat was protective against mild famine we tend to have a higher BMI under a “normal” diet (skip recent US trends, were going through a transition period and haven’t figured out how to work it yet) as it’s more advantageous for most circumstances in the wild.
At the same time, a high BMI can largely be an indicator of co-morbidities itself. As a pure anecdote (myself): I gained more than a hundred pounds in a year in my late twenties, at the same time that both my testosterone fell by more than an order of magnitude and I suffered a spinal cord injury. So my then-high BMI was caused by other ailments, rather than obesity causing them – the causal arrows are pointing backwards in this case. Many (I don’t know what proportion) people who are obese with diabetes have a similar story – the early pancreas (and often thyroid too) led to the weight gain which led to (male) hypogonadism which led to obesity which led to insulin resistance which led to pancreatic failure – loop that a few times and you’re right into a terrible spot that’s not readily recoverable. On the their hand all of these are readily treatable and readily identifiable, should the medical system choose to investigate.
I do a lot of consulting with physicians, so I advise as a practical matter that they test all of their obese males for free testosterone, total T, LH, FSH, T3, T4, SHBG, and E2 because they’ll find quite regularly that this is the root of their patients problems. I would expect to see that at very low BMI too, though I don’t recall any good studies on it, which may be a key driver of low BMI mortality (to the extent I’m wrong about the data being dirty). This works a little different in women, but the same hormones matter, you also have to account for menstrual cycle as well so multiple tests are also needed.
I’m pretty sure that very few of your readers are of indeterminate sex.
No tumtums reading it?
Great post! This is by far my favorite of the ACC posts so far.
Wow! This was great!
Veriasium just make a tangentially related video
https://www.youtube.com/watch?v=QRt7LjqJ45k
Thanks for the review!!
Very interesting.
About the ~10% increase in life expectancy: is it possible that it is exactly paralleled by an equivalent reduction in cognitive (or physical) ability, so that the total cognition in life remains constant?
I mean, many mechanical systems may work like this, e.g. cars, having life expectancy that is to a large extent determined by mileage. Maybe a reasonable prior is that biological systems work the same way, with CR prompting a “low-power” mode.
If true, I think this adds an important aspect to the cost-benefit analysis of CR. It suggests that the fun option is also “optimal”, and there is no reason to feel guilty for taking it.
I feel like the collab already addresses this to some degree by pointing out caloric reduction is often really unpleasant to maintain. (I reckon the unpleasantness necessarily eats up some mental resources, or it wouldn’t be noticed.)
That said, even if your observation is true and you value total cognition in life, you may still want to do it. Example: If you think more life will put you into a time where humanity has fixed (and is able to reverse) ageing whereas you think that your regular life is much less likely to hit that sweet spot, it’s worth doing. From that perspective, you’d be trading a couple of decades of an unpleasant life for much more than the extra years gleaned directly from the effort. (You can also chain this logic – a few unpleasant decades now to -> reach a time where advances to technology give you a few more unpleasant decades to -> reach another time where you gain a few more unpleasant decades to -> finally reach a time where you, with hard work, can begin to reverse the effects of age, and then eventually.)
(Note: This isn’t to say you should think like that. It heavily depends on whether you think this is achievable in the respective timespans. But in abstract, it’s a reason you might want to.)
Edit: Although I’d say you shouldn’t feel “guilty” in any case. You’re almost surely not damaging anyone by doing or not doing CR.
Overall this was a great writeup, however, I found it odd that in the section discussing BMI, you cite only the upper half of the range as the cutoff from the meta-analysis:
Per the study, the lowest mortality was the BMI *range* of 22.5-25. That seems important to point out, lest people come away with the impression that a BMI of 23 is somehow unhealthily skinny.
This isn’t meant as criticism in any way (and thank you two for the collaborative effort!), since it would be some degree of unreasonable to expect this tangent to have been pursued, but I’m left to wonder whether the kind of calories make any difference, as some literature suggests. I don’t mean that in the sense of “balanced diet” versus “ad-lib”, but in the sense of e.g. “try to get most of your calories out of carbohydrates, avoid fats” or “try to get most of your calories out of fats, avoid carbohydrates”.
If either of the authors found something about that by the wayside, I’d be curious to hear about it. (But please really only if you happen to have it lying around.)
My one broad criticism of these first two collabs is that I would like to see utility calculations. Yes, they will vary greatly by individual but it gives a starting point. By that I mean – okay, you will live X more years, but your utility for your whole life will drop by Y because of calorie restriction, so is it worth it? (More straight ahead for this one than the meat-eating collaboration, since that’s mostly about the utility of the beings you’re eating.) For this one, they kind of do give you the answer that it’s not worth it, but if they “showed their work” to the same degree as for the rest of the analysis, even though it’s necessarily a back-of-the-envelope calculation, I think that would be very helpful.
Also – these collaborations are excellent, and (what Scott probably intended) models for how people with opposing viewpoints and who actually care about the truth should interact to the benefit of everyone, including themselves.
> Normative Diet (ND) – a diet that is balanced and prevents the onset of obesity. The general analog to USDA’s 2000 calorie diet. (Our term and not in common usage)
This is dangerous misinformation and I think it’s important that it be edited and corrected. 2000 calories is not any sort of recommendation, from the USDA or otherwise, and for the supermajority of people who read this, 2000 calories *is* calorie restriction. Subtracting 30% from that would be an unsustainable weight-loss diet. https://manytools.org/handy/bmr-calculator/go
Thanks for pointing this out.
The wisecracks were devastatingly funny. Bravo! (And bravo on this ponderous but thorough tome!)
One question; you commented that CR in animals causes sterility, and on that basis asserted that CR isn’t adaptive. Did you mean that temporary CR causes lasting, even chronic infertility or that the animals were infertile during the CR period? If it’s the latter, it’s easy to imagine that it’s actually highly adaptive; not only allowing the animal to survive in times and places with insufficient food but also preventing the animals from having unsupportable offspring during that time.
Good point! I think they meant that a lifestyle strategy of calorie reduction would not be a successful, and hence selected, evolutionary strategy.
But as a temporary reaction to decreased food, it makes some sense. I question whether it would be in fact highly adaptive, though. The other animals that didn’t have this decreased fertility would reproduce just as much, and free-ride on the advantage gained by the one that did, granting it no particular advantage compared to them.
Further, in animals with long gestation periods, environmental conditions might have changed by the time the off-spring sprang off.
But then, maybe that’s an explanation to the first objection? It’s better to delay reproduction and be ready once a time of plenty comes around (like in a season or two) than to have a pregnancy (or create an egg or whatever) during lean times and waste the resources on a sickly child.
Combined with syrrim’s speculations, it makes for an interesting story.
Good question! As a human, I’d be perfectly okay with being infertile for my entire life minus ~2 years to have a child.
My interpretation of the yeast situation would be that the yeast behave like state machines, which die when they reach their stopping condition. They naturally live longer when fed less energy, because they progress through states slower. But what makes a yeast life worth living is presumably the states in particular it passes through, not the length of time it spends alive, such that prolonging those states is not a net improvement.
Humans are different in that our metabolism can’t be slowed down arbitrarily, states need to be progressed through at at least a certain rate. Furthermore we can distinguish between the states progressed through merely to stay alive, and those also worthwhile in themselves (the former would amount to involuntary action like the beating of ones heart, the latter would include such necessary action as gathering food, which can be done better or worse). Assuming, as with the yeast, a person has a fixed number of states they will progress through before dying, this implies that a shorter life is actually better lived. We are interested in maximizing the proportion of those states worthwhile for themselves, but the states expended on merely staying alive are linear in time lived. This is borne out in the Minnesota experiment; whereas the participants might have experience a month of extra life, by the account here they lost 6 months of life in their youth.
The main motivation of the model I describe is conservation of energy, such that an increase in calories has to go somewhere, which ends up being towards a more worthwhile life. Naturally, if you eat more and don’t do anything more, then the calories – and therefore the states – are wasted. The place the calories are going in that case is towards an increase in weight, which in turn raises the baseline calories needed merely to survive. So just saying a shorter life is better lived is a simplification, your weight should be stable and healthy. But assuming that, you can evaluate how much you are getting out of life based on the number of calories you consume (more is better).
Childhood caloric restriction could be potentially worthwhile, if (a) it implies fewer states are required merely to survive (the heart needs to pump less blood to move it around the body, less cell divisions need to occur implying less mutation load is accrued), and (b) it does not lead to living a less worthwhile life. (a) Seems to be correct, based on brief googling. (b) I originally thought would be true, since physical size is less important in a world of reduced physical combat and labour. But reduced childhood nutrition also implies a reduction in IQ, which I think makes (b) obviously wrong. The worthiness of a life is therefore roughly invariant over (non-extreme) childhood nutrition.
I’m somewhat disappointed that the potential to trigger an eating disorder was not better highlighted as a potential complication. Eating disorders are one of the deadliest mental illnesses, and I think any non-necessary practice that could trigger them should be under the highest scrutiny. Yes, for many people it won’t matter, but given the severe consequences of eating disorders it seems like something that deserved more focus.
This is, admittedly, at least in part because my own eating disorder was partially triggered by these sorts of “eat nothing! live longer!” ideas.
I thought the same thing.
I’d like to see this addressed too, but I’m not sure we know enough about how eating disorders are caused to answer this. And “lite” eating-disordered behavior is probably…fine. Some people certainly do spend their entire lives eating abnormally without ever getting to a life-threatening level of anorexia (see: every upper-middle-class white woman).
So far I think this is the best AC of the last two years, bravo. Totally speculative evolutionary theory to follow.
The calorie restriction results pairs really nicely with the Grandmother Hypothesis, basically the idea that human lifespans are “tuned” so that child rearing/knowledge transfer can happen between a generation gap of two, up until the grandchildren reach sexual maturity and can start having families of their own. After that point, you’re a mostly useless food drain and your family survives better if you’re gone. But if your own childbirth is pushed back due to low food availability (which seems to be the reason for CR causing infertility – you don’t want to commit to spending energy growing & feeding a child if you don’t have the energy resources to do so), you want to correspondingly push back your own death so that you can be there for your now-delayed grandchildren. And since your children might similarly have delayed childbirth in low-food situations, it makes sense to use your own calorie intake as a gauge of how much to delay your death over your entire lifespan, not just during the timeframe when you are having children of your own.
If this is true, here is a testable hypothesis: CR should not confer any benefits in the last ~13 years of life, since by that point your grandchildren are already born and there is no benefit to further delays. (Perhaps not true if CR delays sexual development? I don’t know if that’s the case.)
According to the article, CR extends lifespan on a large variety of organisms and most of the evidence is for nonhuman organisms. These organisms have a variety of different life strategies. The explanation for how CR works cannot be an evolution adaption that’s specific to the human life strategy.
Really interesting!
How does exercise fit in? If the rate-of-living hypothesis is true, shouldn’t high-calorie-eating athletes like swimmers have ridiculously short lifespans?
ETA: Quick googling suggests sump wresters eat 20,000 calories per day, typically start they’re careers at 15 and end in mid 20s to early 30s. Their life expectancy is definitely shortened, to about 65, but rate-of-living would suggest they wouldn’t even survive their careers.
I thought of this as well. In fact, there is evidence that physical activity “squares the curve” for life expectancy. Athletes have a much reduced chance of dying early, but also a much reduced chance of living very long.
I was looking for this thread to make sure I didn’t post the same question. I’m skeptical of the nice-sounding hypothesis that has major exceptions and doesn’t fit with everyday experiences and expectations. Indeed, it seems to suggest that the optimal strategy for longevity would be to sit around doing nothing and eating very little, which would be predicted to help you live much longer than someone who stays fit and active.
The other strange hypothesis we should come to from the finite-cellular-calorie-capacity idea is that additional calorie consumption increases metabolic rate, which isn’t necessarily the case. Indeed, most obese people have the problem that they’re tired all the time because they experience decreased metabolic activity and excess calorie storage. The problem isn’t that they’re using all those calories they consume, but exactly the opposite – they’re storing all those calories and the excess stores become the problem that ends up shortening their lifespans.
The alternate explanation for most of the results presented is, “being overweight is contraindicated with longevity”. That doesn’t take any major hypothetical leaps, and it doesn’t require us to believe things that are contrary to the evidence. I was surprised this point wasn’t hit harder, especially when comparing populations with less obesity, like Japan, with those that have more obesity, like the USA. It was the 20,000 pound elephant in the room.
Well, I agree with you that the simple finite-cellular-calorie-capacity idea is very likely wrong as it doesn’t seem to pass the smell test. But perhaps in some form it is a real thing.
It’s much easier to generate a hypothesis that sounds correct than it is to generate a hypothesis that is correct. Whenever a hear a hypothesis that makes so much sense that it has to be true I remind myself to be extra cautious.
What about cells passaged in culture? Wouldn’t different cell types be more susceptible to aging than others? Smells rotten to me, and I don’t see good prospective studies testing its validity.
This is a pretty good write-up of the matter. I applaud you both.
None of these sound good, though, assuming that you do not restrict the type of food consumed. Major problem being that the USDA has no clue how to prevent obesity in practice, if they did, they would be effective at this task.
I like things written in formal academic language; I like things written in casual language; I don’t think I like both welded together inorganically. The “lmao” etc does not add anything to your impressive contribution, imo.
That’s how I felt as well.
Eh, this was fine to my eyes/ears. Compare/contrast the vegetarianism one, where there were some jokes that felt very out of place when inserted in discussions of how animals live horrible lives of constant torture.
I enjoyed it, precisely because it was so ridiculously out of place – to me it fit with some of the jokes (1.5 X chromosome reader) and puns. Though I’m not honestly trying to defend it, since despite my great enjoyment of the abrupt and more noticeable stylistic break, I don’t think the article would suffer from it being removed. 🙂
I also thought this, but then I thought that Scott mixes formal and informal language to great effect in his posts… Maybe it takes a lot of skill to do well.
I’m quite curious on the variance in outcomes for means of calorie reduction. For instance, consider the following situations where people reduce their 2000-calorie diets by 5%:
1. Adam eats a slightly smaller meal portions than the average guy. 1900 calories rather than 2000 daily.
2. Bob skips dinner once a week. Dinner being a larger meal than the others, this also means he’s averaging 1900 a day.
3. Charlie fasts for two weeks straight every year. He just drinks water. Post fast, he eases back into regular 2000-calorie sized portions, so he also has a 1900-calorie reduction.
I unfoundedly speculate that Charlie will have the greatest health benefits! How to actually study this given the lack of Charlies that are comparable in other ways to the rest of the population?
+10 points for “it is not unreasonable to assume that the average American BMI is roughly 30 based on [a] huge body of evidence.”
ugh, i just got that one.
I’m trying to wrap my head around this. My prior is that a BMI of 25 would be the maximum weight recommended, and that optimal weight is somewhere below 25. Current recommendations are that BMI should be in the 18.5-25 range, and I generally aim for around the 19-20 level. Does this result indicate that I should instead be aiming to keep my BMI pretty much exactly at 25? Just for context, for me that would mean I would need to gain approximately 30 pounds.
I’m not quite sure why, but this feels like a cop-out. If it’s not feasible to be on CR without malnutrition, then shouldn’t that just default you into not recommended CR?
It’s very feasible, the point is that people don’t do it.
Is it?
Presumably everyone who is dieting or on some kind of long term CR lifestyle wants to avoid malnutrition and is making some kind of effort to get a balanced nutrient load. The kind of person I know who’s able to keep to, say 1700 or 2200 calories a day isn’t likely to just eat potato chips and soda for their daily requisite; they’re off eating kale and beans and other healthy, supposedly nutritious foods.
If you’re claiming that all these people aren’t, in fact, getting their proper nutrients despite being preselected from the population for impulse control and proclivity towards overall healthier eating habits, then I’d say that’s evidence that CR isn’t feasible.
I have in fact done CR(ON) and there are others who do as well (very few) and if you are tracking your nutrients its very possible to hit all your targets every day in 1500 calories. It’s not EASY though- you have to keep track and plan it pretty well. After a long enough time it gets easier, but I can easily see people like those commenters above who skip meals not hitting their targets.
“A CR diet is reduced calorie intake without malnutrition. Some of the people with BMIs below 25 may have an underlying health issue, such as HIV, which is causing their BMI to drop or mortality to rise, but I speculate that most people with a BMI below 25 are suffering from malnutrition, which is causing the increased mortality observed in persons with BMI below 24-25.”
I think this is completely absurd, “most people with a BMI below 25 are suffering from malnutrition”? (I have a BMI of around 21, 1m75 and 65kg. I do exercise everyday but I eat well and pretty much everything, including the occasional pair of bigmacs after a night out).
*Edit: of course, people are different, but still.. A BMI of 25 for height 1m75 corresponds to 77Kg. (Most 1m75 people weighing less than 77Kg are suffering from malnutrition?)
Some health experts complain that overweight people often suffer from malnutrition, meaning that they get too much energy, but still not enough of some specific nutrients. So it’s possible that also many normal-weight people have malnutrition, but we don’t know how many and how this maps to BMI.
This is a statistic describing a (presumably) normally-distributed population. It’s not a blanket statement about a magical number below which all people are malnourished. Naturally, your individual experience may not be described by the statistic any more than the statistic has to describe your individual experience.
Could the muscular athletes for whom BMI is a bad measure (as a thread above notes) be skewing this “BMI of 25 is the healthiest” statistic?
Seems almost impossible that there would be enough to cause a skew.
I thought this was quite a lot stronger than the previous two, although (especially for the vegetarianism one) that’s probably in part down to choosing a fairly well-defined question. I liked both the citations and the informal style.
Comments:
It seems very strange to conclude that a 10% increase in lifespan would be a nothingburger. That isn’t far away from what you get from quitting smoking!
I’d have liked more focus on moderate calorie restrictions. It sounds like 25% restriction is basically anorexia, in which case it seems like an obviously bad idea even if it has lifespan benefits. But my impression is that most CR advocates suggest something much more moderate (maybe 5-10%?).
AFAIK, BMI is a bad measure for many things, e.g. healthy muscular people often have “overweight” BMIs. I don’t know how much muscle you need to have before this starts being a factor, but if it’s sufficiently low enough that most people who do exercise (that isn’t e.g. marathon running) have “artificially high” BMIs then this seems like it could explain why underweight people tend to have lower lifespans.
I agree with your compliments.
I don’t think that’s quite their view.
They said it was modest, and not recommended in light of the drawbacks of potential loss of focus, resiliency, etc.
Famously Evander Holyfield was considered obese by BMI when he was at his physical prime, when he looked like this.
Yes, BMI is bad. Waist-to-height ratio is better, but still shows slightly increased mortality in underweight. What I’ve seen done once is taking the highest lifetime BMI, rather than BMI itself. Guyenet does this here.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4157748/
Great write-up, very clear, very informative , very funny, thanks for a great text!
I want to emphasize that implementing actual calorie restriction (by 5, 10 or 20%) is impossible outside controlled laboratory conditions. There is no point whatsoever to recommending that average, non-imprisoned Americans with free will and average understanding of nutrition follow a “just reduce your calories by 20% from what you ate before/want to eat!” diet in order to lose weight or become healthier, because it is impossible.
For the following reasons:
– your body changes hunger signals based on how much you give it, and you don’t realize it
– your body changes your level of activity based on how much you give it (and when, and what) and you don’t realize it
– not all calories are the same, and the average American doesn’t know this, and most don’t receive information otherwise, and your body will change hunger signals based on that, and again you don’t realize it
– calorie counts on foods are not always accurate, and for the “good” home-cooked, non processed foods that people are recommended, even less accurate or entirely unavailable. How many calories of broccoli did I put into this dish?
– “Calorie reduction by 20%” always implies accurately knowing what amount of calories you were eating before, and unless you spend a lot of time tracking specific calories, this actually translates to “instead of eating 4 donuts, eat 3 donuts.” But how do you know that “before” you were gonna eat 4?
– almost no one has the time or willpower or expertise to track their actual calorie intake on an individual level
– almost no one has the time or willpower to even accurately track the food they are eating, because on some level we know its pointless and you can’t make people do things they know are pointless, and if you try they will just lie about doing it and totally mess up your data
If you want to recommend diet interventions that average Americans can actually do to improve their health, they have to be things they can actually measure, analyze and do, such as:
– don’t eat food X (soda, etc…)
– eat more food Y (veggies, etc…)
– don’t eat anything after dinner (pre-sleep intermittent fasting)
– don’t eat anything in the morning until lunch (post-sleep IF)
Even the big “is fat or carbs bad?” debate is almost entirely pointless for normal people because normal people do not know how many carbs or how much fat is in the things they are actually eating, or what “high” or “low” fat or carb levels are, or the differences (if there even are any!) between “good” or “bad” carbs or fat. The most important items (good and bad) in the American diet is not actually that wide-ranging. Don’t tell people “cut your carbs/fat” tell them to stop BUYING Item(s) X (soda, chips, candy, snack bars, donuts, etc..) that are the worst offenders, and suggest replacement items (that aren’t terribly unhealthy) that fit the same culinary niche. Because people aren’t buying these things because they’re stupid, they’re buying them because they taste great. Diet programs that rely entirely on ascetic willpower will only work for people who have ascetic willpower, and those people are way more likely to do fine on their own. This kind of selection bias is extremely damaging to almost all diet studies.
Agreed. Have you read The Hungry Brain by any chance?
It’s not impossible. I do it. All the reasons you give for why it can’t possibly work belie the fact that it does work.
It definitely requires a special type of phenotype, but it’s quite rare, especially in the people who have a weight problem.
That’s definitely possible.
I wouldn’t want to discourage someone from trying this if they felt their weight was getting high though- which I think the top comment is saying, don’t bother. Get a scale, get Cronometer, give it a try. It really takes about 10 minutes a day for me and involves no deprivation.
Not sure what’s “impossible” about it. Using a calorie-counting app and a 1500 calorie deficit, I’m down 34 lbs in 73 days.
For me, I think it comes down to a few things:
* The calorie counting forces you to actually consider whether you are actually hungry or just bored (i.e. less mindless eating).
* You really get to know which foods are more filling at a particular calorie count (e.g. 5 eggs vs 1 packet of Ramen).
* You very quickly learn the importance of not “drinking your calories”.
* In my case, I enjoy the game aspect of setting a difficult budget and then trying to beat it.
I did this for a couple of months once. I think the biggest factor in limiting my food intake was thinking “ugh, do I really want to weigh and log that right now?”
Although the process did help me calibrate my intuitions about nutrition a bit, I have very low confidence in the actual calorie counting, because even if you log most meals religiously and weigh every ingredient, the times when you can’t log it accurately (someone else cooked, restaurant food, etc) are likely to be large sources of calories.
The authors seem to be conflating caloric restriction and obesity. They are completely different things, and all that stuff connecting BMI to mortality is irrelevant. A manual laborer or an athlete would quickly die with 2000 calories per day. Consider an athlete who eats 5000kcal per day: is their high-calorie diet killing them or are they fine because of their low bf%?
You are making an excellent point, and your athlete example is great – that our activity level matters. And what’s worse, our calorie intake/diet and activity levels are NOT independent of each other. If that olympic athlete somehow magically adopted that “20% calorie restriction,” their activity levels (and also, calories burned during that activity) would necessarily fall (no matter how disciplined a trainer they were) as their body acclimated itself to an expected lower calorie intake. The athlete’s body (and your body, perhaps even moreso because you probably have an “ad libitum” activity level, unlike them, who is probably forcing themselves to train each day) is not a black box that accepts X calories and produces Y level of obesity. Acting like it is (as this study does) is going to lead you to error.
Just a reminder that most people aren’t Olympic Athletes, and this kind of advice isn’t really directed at that kind of person. If you are an avid runner putting in 30-40 miles per week, a lot of advice that applies to the average American is not going to apply to you.
Peanut butter is not a good dietary recommendation for people with peanut allergies. Milk is not a good dietary recommendation for people who are lactose intolerant. That there are edge cases doesn’t mean you throw out what makes sense for the vast majority, where the advice/information is targeted.
This seems…self-evident, yet we have to go through this every time anything about health/diet/activity levels comes up.
I mean the topic they chose is clearly “caloric restriction”, not “is having a healthy weight good for you”. It doesn’t matter who it’s relevant to.
It’s obvious that the avid runner shouldn’t cut their calories below 2000 while maintaining their running schedule. It’s not at all obvious, based on this post, whether they should give up running so that they can reduce their calorie intake.
As I understand it, if you’re doing CR, you don’t get to be a serious athlete.
Hi guys! Thank you for a wonderful collaboration.
I’m curious what advice you’d now give to BMI-underweight people whose ad-lib is a lot fewer than 2000 calories a day. If a person’s unconscious default would be to often miss a few meals in a row, should such a person force themselves to eat to ensure they eat closer to a normative diet, act according to ad-lib, or reduce intake below ad-lib?
I’d also be interested in this!
(I can currently be observed adding butter to my meals to get my calories up.)
The confusing results wrt the Okinawa longevity stuff might be because the effect isn’t real.
Questioned very recently “Supercentenarians and the oldest-old are concentrated into regions with no birth certificates and short lifespans”
https://www.biorxiv.org/content/10.1101/704080v1
How true! Also, some of those people were counted as centenarians because their families hid their deaths — they wanted to keep collecting the deceased’s pensions!
I’m loving the additional information from the readers at the bottom of these.
Makes me think that they should be in a wiki format of some kind, or go through multiple revisions, with the comments from readers taken into account.
I can’t help but think that the BMI is a useless metric – muscle / fat distribution have to be taken into account.
Muscle/fat distribution only have to be taken into account when they are outside the bounds of normality as defined by the unadjusted BMI scale[1]. Everyone is already aware of your point, but it just isn’t relevant most of the time.
[1] That is, we have a BMI scale which makes judgments without taking body % muscle mass into account, and “normality” as defined by the scale is any level of % muscle mass for which the scale’s labels are not misleading.
Aside from muscle mass, the definition of BMI only uses one dimension, height. People vary quite a lot in how wide they are, whether or not overweight.
I would think a definition that used some measure of skeletal width as well would be more useful, but presumably whoever invented the BMI was worried that that would make it easier for people to fudge the measurement in order to avoid recognizing that they were overweight.
So include waist circumference and body fat percentage, too. But most people who are overfat by BMI will be overfat by those measurements too.
This is an excellent biology joke, but Arabidopsis thaliana is still insulted.
This comment is an excellent and underrated biology joke in itself.
Entertaining write-up! This:
What I’ve seen for this suggests that the “paradox” is best explained by reverse causality due to a selection bias for being underweight at the study period to be associated with illness. The best evidence for this is that measuring “max BMI” does find a positive association with increased mortality. This seems quite germane to the essay since much of the argument against CR in humans is based on this correlational epidemiological finding that in my opinion probably best explained by selection bias. This is mentioned as a possibility but the evidence for it is not presented as far as I can tell. More here: http://ldi.upenn.edu/voices/2014/08/07/explaining-the-obesity-paradox
“Claim 0: ‘You, the reader, should adopt an ad-lib diet.’ Nobody claims this, put the chips down!”
Uh, quite a lot of people claim this.
Firstly, even people who THINK they’re counting calories, whether successfully or unsuccessfully, are usually bending the truth a lot more than they realise, so really they’re going by instinct.
Secondly, most people DON’T count calories. They eat what feels healthy. Or eat what they feel like, but if it feels like too much, cut back. Or just whatever.
Thirdly, “what you feel like” isn’t the same as “whahey, lots”. There’s lots of pleasures where too much isn’t really pleasurable any more, and not everyone is driven to have as much as possible.
Fourthly, what on earth is “as much as you want minus 30%”? How do you measure “how much you want” if you don’t reach it? How do you know when you’re GOING to feel full in time to stop short?
Fifthly, lots of biologists think that whether someone has health problems caused by or co-caused with having too high a weight, forcibly cutting back against whatever homeostasis system current rules just makes it push back harder, so eating what comes naturally is safer than cutting back.
Sixthly, a lot of people think that even if your goal is to have anyone who “looks” or feels overweight to diet harder, mocking and belittling them harms more people than it helps, from feeling hopeless, and developing depression and eating disorders. I guess technically that’s not an argument against your position, just your humour.
I know lots of people DON’T agree. But “eating less is just a matter of willpower and will make people go to a chosen weight” is itself very contentious, leading in with “haha fat people are stupid and lazy” without any actual evidence is not a good basis for the rest of the argument.
> Fourthly, what on earth is “as much as you want minus 30%”? How do you measure “how much you want” if you don’t reach it? How do you know when you’re GOING to feel full in time to stop short?
First, you figure out how much ‘as much as you want’ is by eating until you stop wanting to eat and keeping careful track of how much food that turns out to be on a daily and weekly basis. Then, you work out a diet which contains 70% as many calories (but just as many nutrients) on a daily and weekly basis and eat that for a few weeks. Eventually, you develop an intuition for how much food ‘70%’ is and can keep to that much without precise measurements.
That’s not to say that you *should* do this (the ACC and common sense both conclude that going on a semi-starvation diet without strong evidence of it being a good idea is, in fact, a bad idea), but you could do it if you wanted to.
(Incidentally, does anyone have a good syntax guide for wordpress comment formatting? The wordpress documentation page is a hot mess.)
I think it’s easier than that. Learn to differentiate between hunger and impulses to enjoy something tasty. Learn to stop eating when satiated rather than stuffed or with a clean plate.
The will power isn’t trivial, but I think you can get most of the way there without any bookkeeping.
Almost no American is capable of doing this accurately, and I include myself (and most doctors, nutrition experts and dieticians) in this category – not because we’re lazy or stupid or gluttonous, but because:
– it is extremely difficult to actually track that for many hard scientific reasons
– the implication is that if you properly measured and implemented this 30% reduction, you would see some correspondingly-easily-measured reduction in your obesity, (or increase in health) which you won’t, because there are many other factors in play that are affected by chronic calorie restriction that you aren’t measuring
– it is a gigantic waste of time to do it this way, and your body/brain will very rapidly realize that, and radically reduce the amount of motivation you have to do it
Where is your study? Or, speak for yourself?
I have no problem figuring out how much I have eaten, I use a scale and a simple app Cronometer. It takes an extremely minimal amount of time. I have gained and lost weight (on purpose) many times in my life and it has always been quite simple. Eat more = gain weight. Eat less = lose weight.
I’m not saying calories in/calories out is all there is, but it is a pretty good model for most people. Eat less and your weight will go down.
BTW if you track your food and see what nutrients are getting, it may cause you to eat a bit healthier food as well.
For most people, this must seem like torture. Otherwise, why would all these trials to cure obesity fail? Why do we still have an obesity problem? “Eat less, move more” is standard mantra, after all. You can’t claim people don’t know this method of weight loss. You can’t also claim that they wouldn’t do it if it were simple and easy; AFAIK, they actually attempt it all the time, and in the vast majority of cases fail after a brief, temporary success.
https://www.ncbi.nlm.nih.gov/pubmed/16391215
https://sci-hub.se/10.1001/archinte.1959.00270010085011
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4539812/
Well, YMMV. My point is that it’s hardly a universal conclusion that this is an impossible thing for humans. How could it be? Weighing your food and noting the amount in an app is impossible? Clearly not.
My biggest problem is with this:
What’s the evidence for that? Am I really the only freak who loses weight when cutting caloric intake by 30%?
The trick is not whether cutting calories loses you weight. It does. But the adiposity regulation system will severely cut your BMR if you lose enough weight. So you would progressively need to keep cutting to keep losing. The collaboration authors went into depth about the Minnesota Starvation Experiment, but here’s some newer data, from metabolic measurements of The Biggest Loser contestants. IIRC (can’t recall where I found that out), one poor guy went from a BMR of 3500 kcal to something like 1700 kcal.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3387402/
https://www.ncbi.nlm.nih.gov/pubmed/27136388
>I have no problem figuring out how much I have eaten, I use a scale and a simple app Cronometer. It takes an extremely minimal amount of time.
I tracked everything I ate for three days, and it was a huge headache. Weighing/measuring EVERY ingredient? Even with homemade food? Even with restaurant food? Very difficult with anything that has more than half a dozen ingredients. And I guess you’re bringing a scale or measuring cups to work and restaurants and parties?
You have to either eat a very simple, restricted diet or else accept some very inaccurate calorie estimates.
Yep.
Any long-term successful real-world intervention has to be a) simple, b) ad libitum intake.
I agree completely that the glib comment was inappropriate and counterproductive, but I wouldn’t let it color your perception of the rest of the essay too harshly, since overall it is quite good.
This is hard for me to evaluate — “lots of biologists” isn’t quite a citation. By my (not at all an expert) understanding, the modern obesity epidemic is to a large extent caused by increased access to high-energy density foods, an environment we did not evolve to handle. In other words, our natural homeostatic mechanisms are the problem here, because they’re not suited to the environment we currently live in. Assuming for the moment that your statement is true, though, it’s plausible that more targeted interventions are needed when there are specific health concerns or to deal with acute obesity, while simply maintaining a healthy body weight could be accomplished by the normative diet (as the citations in the essay show).
A lot of people do this. Not a lot of people (probably rounds down to zero) advocate it as a healthy lifestyle, which I’m pretty sure was the point in context (I think it was fairly clear).
Which part did you feel was overly insensitive to overweight people?
I’m not a lot of people, but I think “eat as much as you want” can be a healthy lifestyle. Just not if paired with “eat whatever you want.”
I think that if you cut away the junk, metaboloically healthy people can use their hunger as a guideline to how much they eat.
I tell my kids, “I don’t care what’s left on your plate. Eat until you are full, then stop.” Easier for some than others, etc.
I interpreted ad-lib to mean “as much as you want of whatever you want” (the term ad-lib kind of implies arbitrariness on both ends to me).
Also, “eat as much as you want” doesn’t necessarily end when you are full. Your points are all correct, but unrelated to advocating for the “ad-lib” diet as identified in the paper and referenced in the post I was responding to.
Okay then. I interpreted it more as “eat til you don’t feel hungry anymore.”
What’s the question we’re trying to answer (in this sub-thread)? Can one achieve a healthy diet without careful mental effort and without continual feelings of hunger? Yes, imo.
Can one achieve a healthy diet if one satisfies every advertising-induced whim or impulse? Absolutely not.
Yeah. I think ultimately the question we’re trying to answer is “what is meant by ad-lib” and any disagreement is stemming from varying interpretations of what that means. I agree with your points, I just disagree that what you are describing is what was meant by “ad-lib” here, which is why I disagree with the top level comment that “actually, people do claim that”.
If your definition is what was intended in the article then yes people totally claim that is a healthy lifestyle (and they are likely correct, for at least some significant portion of the population), I just don’t think it is based on the context and the word choice.
The term generally is applied to laboratory rats given unfettered access to food.
Though I don’t think that really answers the question of what is meant when applying it to people. Consciousness and long-term goals complicate things.
I believe the line in question is “put the chips down!”
Ah. Well, that’s gonna be real hard to get worked up over.
That isn’t even making fun of fat people, just the majority of us who eat some variation of Standard American Diet.
The entire Intuitive Eating movement advocates “eat what you want, as much (or little) as you want” as the healthiest way to eat. It’s a common philosophy found in eating disorder recovery, and is also gaining some traction as a way to work with children who are picky eaters or who have sensory issues.
> Fourthly, what on earth is “as much as you want minus 30%”? How do you measure “how much you want” if you don’t reach it? How do you know when you’re GOING to feel full in time to stop short?
That seems pretty easy to do: count calories for a couple of weeks, then cut back to 30% of that.
It’s less of “contentious” and more “conclusively refuted”. Looking at historical data should be enough to conclude this, but there are actual trials (like the Women’s Health Initiative study) where it’s pretty damn clear that even if you move more and eat less, you will not lose more than an insignificant amount of weight in the long term.
This seems rather unlikely.
https://jamanetwork.com/journals/jama/fullarticle/202138
Look, particularly, at figure 2 and table 2 (you need to click a button to show them in the sidebar). 120 kcal difference between control and intervention, 14% difference in physical activity. Yet, the differences in weight at seven years followup are below 1 kilogram, on average.
I see no changes in exercise- physical activity is the same between intervention and control.
Food consumption is based on survey results, and magically the control group cut their own caloric consumption by 15%.
Meanwhile the intervention cut fat and increased carbs, it didn’t just reduce calories.
The most you could argue is that a 7% reduction in calories leads to a 2.5 lb reduction in weight (~1.5%?) but I think that’s very tenuous.
Clearly it is absurd to say that eating fewer calories and burning more will not change your weight in the long term.
Those are incredibly small changes. I would expect small results. And looking at the data, including the figure you reference, you see exactly that: small but clear results.
The large jump in the first year for the intervention group also suggests something that I already suspect about these kinds of studies: the people in the study actively follow the “intervention” guidelines and live generally healthier lifestyles for the first portion, where it is new, and then gradually regress towards (but maybe not all the way back to) their previous behaviors and lifestyle. This may not show up in self reported diet and activity data because people involved in a study about them adopting a healthier lifestyle will probably overstate the extent to which they are following it when giving the reports (i.e. “oops, forgot to report I ate 4 pieces of pizza and 2 slices of cake at the office party last Tuesday” or “yeah, I’ve been walking 2 miles every day (under breath: but I’ve been so busy I haven’t done it in 2 weeks)”.
In other words, that study shows exactly the opposite of what you are claiming, even before you account for the fact that the Intervention group probably wasn’t as intervened after the first few years as the study data would make you believe, leading to understated results.
My bad about the exercise factor, poor phrasing. What the table shows is that both the intervention and control groups increased their exercise. The intervention group increased it slightly more than the controls (non-significant). Which is all the more interesting, given that the control group gained weight on average.
Fortunately, weight measurements are not by food fantasy questionnaire. They must have actually had a caloric deficit at least initially. You can certainly doubt the control group, but not so much the intervention group
With such a small change, I don’t think it matters. In this context, fat calories and carbohydrate calories are interchangeable.
It’s not absurd, if you know that your caloric intake and current body weight modifies your caloric expenditure. I linked to some severe examples in another subthread.
I don’t consider “less than a kilogram of difference” to be a result worth talking about. Especially given how heavy Americans are.
Regression is certainly a factor. These are free-living people. Which is another strike against this strategy – non-sustainability in real world environments.
If you have any evidence that caloric restriction in free-living populations results in long-term resolution of obesity, I’m all ears. I don’t know of any; everything I’ve seen that uses increased exercise and intentionally reduced calories, fails. I use WHI as a counterexample because it was really big and well-funded and long, not because it was particularly good – if I were designing it, I’d be measuring BMR at the very least, to confirm calorie reports.
You are free to ignore statistics (that you presented!) that counter your bogus, inaccurate, harmful narrative at your leisure. I’m pretty much done here other than to encourage anyone who might have taken any of your bad advice to heart to reconsider.
I’m not sure what is it that you think is wrong about my position, unless you have misunderstood it.
The OP stated
and I replied
I have provided WHI as an example of a study that shows that conscious control of calories and exercise does not cure obesity. You pointed out some valid concerns about the limitations of that study. But it is hardly the only piece of evidence. Have some more!
Treatment for obesity by standard weight loss methods (which are still stuff like this in 2014) typically fails hard:
https://sci-hub.se/10.1001/archinte.1959.00270010085011
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4539812/
https://www.ncbi.nlm.nih.gov/pubmed/29156185
A particularly egregious example are the studies on The Biggest Loser contestants, who despite massive losses, have regained their weight, and their BMR was still in the dumps after 6 years.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3387402/
https://www.ncbi.nlm.nih.gov/pubmed/27136388
This is all because they are treating the wrong problem. Voluntary overeating is not the cause of obesity. Voluntary overeating requires discipline and is not comfortable, and I say that as someone who has done this for experimental reasons. In terms of how pleasant that is, it’s somewhere between prolonged water fasting and prolonged dry fasting. Overfeeding ramps up TEE, which makes weight gain much slower than you would expect from just the extra caloric intake.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6054727/
The unconscious parts of the brain regulate how fat you are supposed to be, which includes parameters like BMR, non-exercise physical activity and interest in food. Trying to use the conscious brain to manage this is like wrestling with a robot who doesn’t tire, and gets stronger the more you start winning. I invite you to check out the references in part 1 of Guyenet’s notes for the debate with Taubes. (Or at least our host’s review of Guyenet’s Hungry Brain.)
I don’t know, like I said I have deliberately gained weight on multiple occasions. At one time I was eating about 5,500 calories per day. I didn’t have much trouble with that. In fact the most annoying thing was how frequently I had to use the bathroom.
It’s really about what kind of food you eat, right? If you only have fruit, vegetables, nuts, fish and water, does any of that stuff about the unconscious parts of your brain matter? I don’t see how it can. Otherwise how can you explain the change over time?
@Cliff
Whereas I experienced nausea three out of the five days I did it on purpose. I was eating well over 6000 kcal per day, using fresh and processed meats, butter and eggs.
Yes, the food you eat matters a great deal. I mean, raw vegans can eat a ton of stuff, and they look like skeletons if they keep it up. The more unrefined the food you eat, the lower the set point, seems to me. Although that is just a heuristic – doesn’t necessarily mean it’s true for all processed foods in all contexts. Certainly not true for something like a Kempnerian Rice Diet (sugar, rice and fruit juices), or Keto Animal Foods (low protein, high fat version of the Carnivore diet), or the Croissant Diet (stearic acid enriched croissants as primary foodstuff).
I really like how you set-up the article by being very precise about your specific opinions and about the claims you want to evaluate, and especially phrasing the claims in a “you should” fashion.
This prevents the two things I hate most when discussing opinions. Many people don’t even know what their opinion actually is and (often as a result) can’t say how that opinion should affect decision making.
I liked the writing style, good mix of funny and informative, full of common sense…
My own summary: slight CR will probably slightly delay your death, but the adverses effects of CR all look exactly like symptoms of old age…
So CR=aging earlier and dying later. Not for me…
Amusing way to put it.
Note that it will only delay your death if you are in a rather sterile industrial nation. If you encounter physical hazards or periodic famine, it seems a very risky strategy.
On the last note: elderly people loosing weight is indeed a bad sign. I remember my grandma, who had been overweight since her forties, suddenly lost weight. She died three months later. Slightly fatter elderly people just look better, also (better skin color, more joyful).
That doesn’t mean obesity is good, but an elderly (70+) person losing weight is usually almost never good, even if they were obese. At least, every time I’ve seen such sudden changes among my family and friends’ family, a funeral followed.
Yeah, death gives you a different perspective on all those pounds we struggle to keep off and why your body wanted them in the first place.
I’m not sure it’s the weight per se that keeps you healthy, or what leads to it. In my grandma’s case, she just lost her joy of food. She liked fried chicken with skin, cakes, sweets, and all kinds of unhealthy food. After she lost weight, it seemed more like she was just pushing food into herself to keep herself alive than because she enjoyed it. She ate chicken breast, veggies, etc, in small quantities.
She died of a heart attack, and AFAIK, heart issues are not connected to appetite, but whatever caused that loss in appetite wasn’t good.
That sounds like end-stage type 2 diabetes – all of the prior obesity, the unexpected weight loss, and the cardiovascular illness.
Careful — “had been overweight since her 40s” doesn’t equate to “prior obesity”. “Overweight” and “obese” are two different weight classes in the BMI x gender space, with radically different health implications.
If I recall correctly, all-cause mortality is lower among the “overweight” than among the “normal weight” group.
No, she wasn’t diabetic. In Russia, they autopsy all deaths, so we knew the cause of death, and that was a heart attack, which in women frequently presents differently than in men.
Is the second sentence supposed to be the basis for the first? Because that definitely does not follow.
@Michael Watts
A fair point. I don’t know how overweight she was. Still, you don’t have to be overweight at all to be diabetic, if your personal fat threshold is low.
@ana53294
On the basis of what was the diagnosis made? High fasting glucose is typical, and a really bad test for diabetes. Oral glucose tolerance tests are a little better, but still bad for diagnosing diabetes. Just about the only test with excellent sensitivity and specificity is a Kraft-type insulin assay. Kraft himself famously stated that everyone with heart disease who isn’t diabetic – is simply undiagnosed. This is probably false on the margins, since there are non-diabetic causes of heart disease, but given the prevalence of occult diabetes in the population, I’m willing to bet that it accounts for most of the heart disease by far.
A doctor friend of mine once claimed “unexplained weight loss in the elderly is cancer until proven otherwise”. (Although I mentioned this to him more recently and I’m not sure he stands by it now.)
Cool collaboration! Write-up was a little rougher around the edges than the others so far, and cited to within an inch of its life, but it was very interesting and informative all the same. Relative to the first two collaborations, it strikes me that evaluating strictly medical / scientific data on a tightly restricted question without having to worry about the ethical foundation is Easy Mode– not necessarily meaning that as a criticism; I appreciate the definiteness it makes possible.
I came away quite persuaded that (1) CR is a promising strategy if one’s goal is purely to extend lifespan, and (2) there’s no free lunch here, because it would make me more than miserable enough to compensate. Fewer revolutions and more food is an understandable preference; I guess nobody asked the lab rats.
Pros:
– Taught me what aging actually is and why / how it’s different from life expectancy
– ALL the citations, endnoted and non-hyperlinked in case I was tempted to actually look
– Seems to have more or less reached an uncontroversial, common-sense right answer
– Liked the fact that the authors made their back-and-forth part of the write-up; it made the reasoning more transparent and easy to follow
Cons:
– Not a hugely high-stakes issue; maybe not even a difficult one
– Walls of stats and citations made my eyes glaze over occasionally
– Writing style was a little inconsistent, though entertaining
In other words, the pros and the cons are more or less the same. Comparing this to, say, the meat-eating collaboration is going to be tough as they are very much apples and oranges.
I rate this collaboration as a 7/10 (no particular scale or judgment implied, this is just for my own reference). As always, many thanks to the authors for putting in the work to create this.
(Disclaimer – Biology PhD student here)
This IS the proper amount of citation for a review of this kind. Other entries (meat eating in particular) felt lacking in this department.
On the post itself – great job. This leaves very little desire to go and double check everything or just disregard it and do your own research – personal indicator for a good review.
Really? But they didn’t cite the sinking of the Lusitania!
(I personally greatly enjoyed the style; if anything, it felt more comfortable/easier to read than the previous ones, though I’ve been enjoying all of them. But I’ve read enough things with this many citations to not find them distracting, and it’s nice to be able to check the source on any individual claim that seems odd.)
See also: https://www.smbc-comics.com/comic/2013-02-06
>Not a hugely high-stakes issue; maybe not even a difficult one
Really? The possibility of extending one’s life by everyday choices seems incredibly high-stakes to me.
I really liked the writing style—good balance of well-cited and entertaining.
Well done! On first read, this seems much less one-sided than the last 2 ACC entries. Intent and voice of both authors clear. Major facts and lines of inquiry are covered with sufficient detail.
Main concern is that it’s not clear how they reached the conclusion’s expected lifespan increase, which seems like a larger effect size than the evidence warrants. Only other things I’d have like to see are more analysis of the limitations of the chosen animal models and more evaluation of the proposed mechanisms.
Just want to throw this out there: it might be that it’s less one-sided because the evidence is less one-sided. In theory, if there is a clear answer to a factual question, then two people who review the literature are supposed to converge on that answer. You cannot draw a straight line from “the final writeup sounds more balanced” to “the authors on both sides did a better job.” (I do agree that this AC is very good.)
For what it’s worth, I didn’t commend this entry because the evidence is less clear but rather because unlike other collaborations it presented the major arguments from both sides that one interested in the topic would bring up. Also as the comment section shows, their interpretation of the data is less contentious.
This cannot actually be the case — this entry concerns a factual question, while the other two entries are explicitly subjective. As such, the concept of evidence doesn’t apply well to the first two entries.
I would say that this entry sounds less one-sided because, discussing a factual question, you don’t really have the option of just choosing to ignore related ideas that you’d rather not discuss.
My read was that it was mostly due to finding the proposed mechanism (oxidized free radicals) likely but the evidence contradictory.
PPS mentions “DR”– what is that? Typo for CR?
Thanks for asking, I thought I’d forgotten the meaning of the acronym over the course of the piece.
Dietary restriction, maybe? As in not just calories, but micronutrients and macronutrient balance?
Doctor here (and someone who has experimented with IF). This is GREAT! Really enjoyable read.
(I have to add – please don’t take aspirin if you’re healthy. No good evidence for prevention of heart disease and increased risk of major bleeding.)
Would love to see analysis of benefits of IF or time restricted feeding above and beyond simple calorie deficit. Many practitioners argue for hormonal benefits ie increased concentration of growth hormone, lipolysis etc
Yes, this is what I came here to add. I use IF as well, and would have loved to see the analysis cover it.
This analysis ignores IF, which is fine! It mentions it, but seems to imply it is the same as calorie restriction, which it is not. The entire point of the “I” in “intermittent fasting” is that the fasting is intermittent, – calorie restriction may or may not occur. In fact, part of the reason I like IF is that it gets at one of the fundamental reasons, as the authors note, that CR is behaviorially difficult – because we are not black boxes that accept X calories per day and produce Y output of energy expenditure and fat accumulation – it way easier to just extend the daily time I don’t eat (no one thinks that having a 14-16 period in your day where you don’t eat is a level of “calorie restriction” that will harm you via “starvation” effects). Simply reducing your calorie consumption by 20% a day is extremely hard to adhere to for many reasons, and your body will respond to it differently than intermittent fasting, whether or not that IF results in total CR of 20% or 10% or even an increase.
There is evidence that the beneficial effects of IF are due to CR even if you are not trying for CR
I was under the impression that some believed IF carried benefits relating to waste disposal, damaged cell apoptosis, insulin sensitivity, lipostat, etc due to activating pathways associated with mild short term deprivation, which would be activated regularly in the EEA even with no CR but which aren’t activated under a Western 3-square-meals-plus-snacks regime of constant anabolism?
Spot on – eg:
https://www.sciencedirect.com/science/article/abs/pii/S095528630400261X
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3106288/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC329619/
I would think that if nothing else, IF is going to exercise your endocrine system, i.e. make sure your liver and other organs are cooperating to deal with variance in the food supply, switching on fat breakdown and such as necessary.
It seems implausible that we evolved to live best on a continuous drip feed of the correct nutrient composition.
I have read that a very regular heartbeat is said to be a harbinger of cardiac trouble (though I admit that a quick google just now has yielded nothing along those lines). Still, it makes sense to shake things up a little in every part of life, surely?
Are you interpreting Intermittent fasting as going for 14-16 hours between your last meal of the day and your first meal of the next day? Is there evidence that that increases life expectancy?
Nice if it’s true, since I already do that.
It’s commonly defined as 16 hour break in eating, yes. But a very common failure mode is to mistake skipping breakfast for IF – if you actually count the hours, you rarely get to 16 hours by just skipping breakfast.
Thanks. I’ve been doing a 16 hour fast for some time now, following the recommendation of Bredesen’s book (The End of Alzheimer’s) for people with one copy of APOE4, hence greater than average risk of developing Alzheimer’s.
Whether or not it works for that, I think it tends to reduce my weight, just by eliminating late night nibbling.
Re: low dose aspirin regime, what about bowel cancer reduction and the tiny, but constantly replicated all cause mortality reduction, under a proposed mechanism of not just blood thinning, but general mild inflammatory reduction? Gwern’s got a tweetstorm and a more thorough analysis on the subject that really convinced me it was worth it for anyone who didn’t have bleeding-related health issues (or family history or other risk factors for such). And especially worth it for people with elevated cancer and/or cardiac risk.
I haven’t read it – how recently was it done? The medical stance on this has changed somewhat over the past year due to papers like this one:
https://www.nejm.org/doi/full/10.1056/NEJMoa1800722?query=featured_home
That’s a pretty big selection bias. Aspirin is supposed to help with cardiovascular disease in particular.
Anyways, when making personal decisions we should really take personal factors into consideration. For example I have about four mild risk factors for stroke (family, migraines and a couple of coagulation related mutations). For me the only decision is which age do I start. Somebody without any risk factors but maybe slower than average coagulation or gastrointestinal problems the decision would be again obvious. For the rest it’s probably just not very important.
This is perfectly consistent with the research results, and makes a lot mores sense on an individual basis.
Calorie restriction shortens lifespan in more mouse strains than it extends it in. In fact the strains that experience life extension appear to be increasingly overweight with age. It should be no surprise that calorie restriction in the context of obesity would promote health, outside of that context there is very little evidence for it.
Carlorie resctrictiion does not extend lifespan in wild derived mice
The primate data pretty clearly did not show life extension in CR animals despite the authors clear preference that it be otherwise. The study that claimed life extension you correctly pointed out how it has major issues to say the least.
Slightly underweight humans have higher mortality that normal weight ones when controling for many confounders (including smoking and disease), slightly overweight humans have the lowest mortality.
CR as a field is largely driven by publication bias and has largely been debunked as a plausible longevity intervention.
Your conclusion that CR is likely to extend lifespan by 10% in humans is totally unsupported by any evidence (including your own review of the primate data), but if it did your further thought that it might not be worth it seems incongruous.
A 10% improvement in life and healthspan would be more time and health than you would get by curing all of atherosclerosis and all cancer combined. People in general appear to be willing to endure incredible hardship for far more modest improvments (take the present treatments for cancer).
The cancer thing does not at all support the theory that people undergo great perils to increase their lifespan. On the contrary, we have overwhelming evidence that people are far less interested in interventions like working out, eating healthy, sleeping enough, and not smoking than they are in spending a lot of money on medical treatments (“far less” as measured by a cost/benefit analysis wrt health and longevity). The punchline here is that people are either completely irrational or optimize for something other than lifespan (such as peer approval). The one thing most humans are definitely not doing is rationally optimizing for longevity.
To your first post, obviously, if you respond to a thorough literature review by citing your own two favorite papers claiming the opposite, this is not convincing. It’s particularly bad if you say stuff like “it’s totally unsupported by any evidence” if the authors just went through tons of evidence.
Unfortunately, this is probably correct, and it’s a shame. I was more concerned that the authors seem to believe in a ~10% survival benefit and yet don’t recommend doing it. I’m glad they don’t recommend this because it’s probably dangerous (those people I know who practice CR all appear very unhealthy and complain of getting URIs quite often).
I’m afraid it doesn’t work that way. There cannot be a thorough literature review of this topic without those two papers, or at least one of them, or otherwise just ignore all rodent data and look only at the primate data. CR as a field has been dying a slow death since those papers were published (the death accelerating since the primate data was released).
CR based life extension in rodents appears to be an artifact of using some specific inbred mouse strains prone to obesity. This is very important to this topic and relevant.
They shared no evidence relevant to non-obese human beings (and it’s non-interesting that obese humans should reduce calories)
No; yeast, worm, and some inbred rodent strains are not relevant. Both yeast and worms literally just enter a type of suspended animation when nutrients are lacking; at the margin they partially enter this state. Some inbred rodent strains benefit from CR, most don’t, those that do tend to be obese.
The primate data is of course the most relevant to humans, and it is firmly on the side of demonstrating a lack of life extension in non-obese animals. In one research center they did a reasonable job with their control animals (they didn’t let them become very obese). In the other they did the equivalent of locking them in a McDonalds and giving them nothing to do with their lives but eat and get fat; this group found that when reducing this intake, it was beneficial. This is obvious, and not interesting.
Again, slightly overweight humans (BMI 25 or 26) have the lowest mortality among all weight classes. Underweight humans have high mortality; even when controlling for smoking and disease (the concern of course is that we can’t control for unknown disease).
Humans practicing CR do affect a variety of biomarkers (biomarkers that normally have low predictive power like cholesterol). Anecdotally they also get sick more often and appear otherwise frail (frailty normally being highly predictive of mortality). There are no well controlled human studies so we really don’t have human data for now.
So, I suppose I partially retract my statement and replace it with this. “CR as a life extension method in non-obese humans remains unsupported; much relevant evidence suggests that it will not work and may be harmful.”
Aging and Age-related disease is my field so I apologize if I come across at all harshly.
CR aside, what do you think of various current projects to slow or reverse aging? Ones I am aware of include:
A team at Harvard medical looking at genetic approaches, and predicting that they will be able to stop aging in dogs in a few years (not very precise detail–an article I read recently).
Supplements that are supposed to produce NAD+ (Elysium).
A recent experiment, involving sixty people for a couple of years and using (I think) two drugs, which wasn’t aimed at age reversal but resulted in biomarkers of age going substantially down–lower at the end of the two years than at the beginning.
Some rodent (mouse or rat, I forget) experiment with a supplement that apparently increased the speed of healing, mimicking the effect of a particular strain of the species, and appeared to have age reversal effects.
I apologize for the vagueness of all this, but as someone in the field you can probably recognize the different projects I have heard of, and may know of others. Does it look as though any of them is likely to produce significant results in the next decade or two?
Did you reach any conclusions about whether ‘total calories consumed’ is the only thing that matters?
For example, is it better to fast for 2 weeks/yr vs. reduce daily consumption by 4%?
I think the general idea is “don’t be fat”. Whichever sustainable route you choose to that destination, is correct enough.
Intermittent fasting is a form of caloric restriction. While I could understand an argument for it based on increased growth hormone levels (released only in a hypoglycemic state, which is what I assume you’re getting at) absent caloric restriction it’s only the caloric restriction aspects which are held to have an impact on lifespan, AFAIK.
Very interesting! I’m particularly glad you explained the subtleties between different kinds of control diets. I think this is the best ACC so far this year. Some explicity QALY calculations would have been nice, but I think the conclusion is still solid.